New developments in the analysis of complex environmental matrices (2015)
Meeting report by Roger Reeve, Graham Mills and Ian Forber
ECG Committee Members
ECG Bulletin July 2015
ECG Committee Members
ECG Bulletin July 2015
Over 80 delegates and speakers attended this one-day meeting held on Friday, 6th February 2015 at the Royal Society of Chemistry, Burlington House, London. The meeting was organised by the Environmental Chemistry Group (Dr Roger Reeve, Mr Ian Forber) and the Separation Science Group (Professor Graham Mills), and aimed to update delegates with recent developments in analytical instrumentation that make it possible to simultaneously analyse numerous pollutants in complex matrices using minimal sample clean-up. There was a focus on analysis of solids, liquids and gases by liquid or gas chromatographic separation (LC or GC) with mass spectrometric (MS) detection, but one nuclear magnetic resonance (NMR) metabolomics investigation was also presented. Laboratory and field analytical techniques were covered, along with the requirements for high sensitivity and specific analyte identification in complex matrices.
There were eight presentations by speakers from UK academic institutions, agencies and industries. A keynote lecture was given by Professor Damià Barceló (CSIC, Barcelona, Spain). Professor Barceló has an international reputation for the analysis of pollutants in water. Professor Graham Mills (University of Portsmouth, UK) opened the meeting, welcoming the delegates and chairing the first five presentations during the morning session. Lunchtime gave opportunities to network and to inspect sponsors’ displays. The afternoon session started with the keynote lecture delivered by Professor Barceló, and was chaired by Dr Roger Reeve (University of Sunderland, UK).
There were eight presentations by speakers from UK academic institutions, agencies and industries. A keynote lecture was given by Professor Damià Barceló (CSIC, Barcelona, Spain). Professor Barceló has an international reputation for the analysis of pollutants in water. Professor Graham Mills (University of Portsmouth, UK) opened the meeting, welcoming the delegates and chairing the first five presentations during the morning session. Lunchtime gave opportunities to network and to inspect sponsors’ displays. The afternoon session started with the keynote lecture delivered by Professor Barceló, and was chaired by Dr Roger Reeve (University of Sunderland, UK).
Abstracts
High resolution mass spectrometry provides novel insights into products of human metabolism of organophosphate and brominated flame retardants Dr Mohamed Abdallah (University of Birmingham, Birmingham, UK)
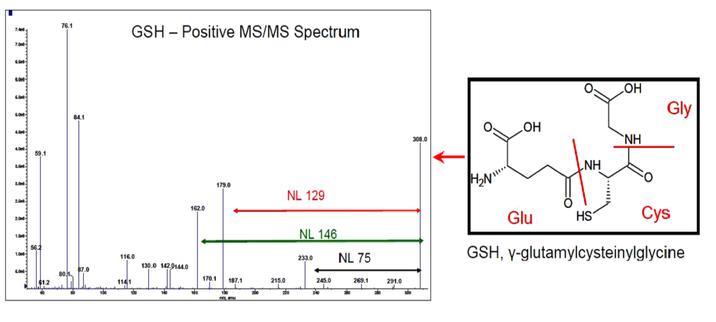
Indoor dust has been consistently identified as a major pathway for human exposure to flame retardants. Few studies have been made on metabolic pathways. Many of the studies investigate the banned polybrominated dibenzofurans (PDBEs), some with alternative retardants and only a few mimicking realistic in vivo situations. The current work studied the metabolism of mixtures of brominated and organophosphate flame retardants applied to human hepatocyte cultures as a dust sample and as a synthetic mixture containing similar concentrations of the retardants. The dosage of the cells was designed to be equivalent to 12 mg dust based on a 12.3 kg toddler ingesting 200 mg dust/day. After 24 hours incubation, the metabolites were extracted with methanol and analysed by ultra-high performance liquid chromatography-electrospray ionisation mass spectroscopy (UPLC-ESI-OrbitrapTM MS). Metabolite identification was by MS full scan: four digit accurate mass with confirmation by MS/MS (Figure 1). Phase I and Phase II metabolism were identified, and all ion fragmentation spectra were useful for identification of the conjugates. Details of the metabolic pathways of each compound type were determined.
Indoor dust has been consistently identified as a major pathway for human exposure to flame retardants. Few studies have been made on metabolic pathways. Many of the studies investigate the banned polybrominated dibenzofurans (PDBEs), some with alternative retardants and only a few mimicking realistic in vivo situations. The current work studied the metabolism of mixtures of brominated and organophosphate flame retardants applied to human hepatocyte cultures as a dust sample and as a synthetic mixture containing similar concentrations of the retardants. The dosage of the cells was designed to be equivalent to 12 mg dust based on a 12.3 kg toddler ingesting 200 mg dust/day. After 24 hours incubation, the metabolites were extracted with methanol and analysed by ultra-high performance liquid chromatography-electrospray ionisation mass spectroscopy (UPLC-ESI-OrbitrapTM MS). Metabolite identification was by MS full scan: four digit accurate mass with confirmation by MS/MS (Figure 1). Phase I and Phase II metabolism were identified, and all ion fragmentation spectra were useful for identification of the conjugates. Details of the metabolic pathways of each compound type were determined.
Use of metabolomics in solving environmental problems Dr Jake Bundy (Imperial College, London, UK)
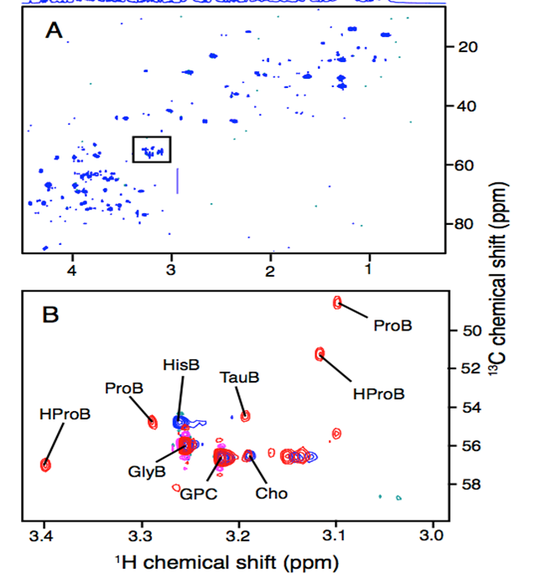
Is it possible to study environmental effects by metabolomic analysis of a single species under a variety of stressors such as pollutants, climatic conditions and feeding levels? Earthworms were studied in the laboratory and at 17 sites (including 6 polluted and 6 control sites) in the UK for which soil properties (TOC, pH, moisture, temperature), and levels of metallic contamination were determined. A total of 300 worms were studied. 1H/13C heteronuclear NMR experiments were undertaken on tissue extracts, restricting data acquired to the ‘interesting’ betaine region to reduce acquisition time (Figure 2). Although it proved possible to link some laboratory stresses to field effects, there was large inter- site and intra-site variation and the model was not predictive. The number of earthworm genotypes makes this difficult (two cryptic species are found in the UK and there are probably several more across Europe). Using this approach, metabolites were identified which could distinguish the different genotypes.
Is it possible to study environmental effects by metabolomic analysis of a single species under a variety of stressors such as pollutants, climatic conditions and feeding levels? Earthworms were studied in the laboratory and at 17 sites (including 6 polluted and 6 control sites) in the UK for which soil properties (TOC, pH, moisture, temperature), and levels of metallic contamination were determined. A total of 300 worms were studied. 1H/13C heteronuclear NMR experiments were undertaken on tissue extracts, restricting data acquired to the ‘interesting’ betaine region to reduce acquisition time (Figure 2). Although it proved possible to link some laboratory stresses to field effects, there was large inter- site and intra-site variation and the model was not predictive. The number of earthworm genotypes makes this difficult (two cryptic species are found in the UK and there are probably several more across Europe). Using this approach, metabolites were identified which could distinguish the different genotypes.
Monitoring of polar pollutants in surface waters using Chemcatcher®-based passive sampling methods Anthony Gravell (Natural Resources Wales, Llanelli Laboratory, Wales)
Passive sampling devices can be deployed directly in rivers for several weeks to give time-weighted average concentrations of pollutants. These avoid the ‘hit and miss’ nature of taking spot samples of water. The acid herbicides monitored were highly water soluble and almost completely ionised at environmental pH, so needed to be extracted by ion-exchange rather than conventional adsorption. Chemcatcher® passive samplers using Empore® anion-exchange discs were deployed in the laboratory and field (10-14 days sampling time) and compared to existing water monitoring methods. Measurements were taken during the linear uptake phase (before equilibrium), where the concentration of pollutant was proportional to the mass of the disc, the sampling rate and the exposure time. For field applications a polyethersulfone membrane was added to the Chemcatcher® to increase the time to reach equilibrium and to improve disc integrity. Typical field concentrations of herbicides were in the low ng/L range. Measurements below the detection limit of the spot sampling method were possible using this novel approach.
Passive sampling devices can be deployed directly in rivers for several weeks to give time-weighted average concentrations of pollutants. These avoid the ‘hit and miss’ nature of taking spot samples of water. The acid herbicides monitored were highly water soluble and almost completely ionised at environmental pH, so needed to be extracted by ion-exchange rather than conventional adsorption. Chemcatcher® passive samplers using Empore® anion-exchange discs were deployed in the laboratory and field (10-14 days sampling time) and compared to existing water monitoring methods. Measurements were taken during the linear uptake phase (before equilibrium), where the concentration of pollutant was proportional to the mass of the disc, the sampling rate and the exposure time. For field applications a polyethersulfone membrane was added to the Chemcatcher® to increase the time to reach equilibrium and to improve disc integrity. Typical field concentrations of herbicides were in the low ng/L range. Measurements below the detection limit of the spot sampling method were possible using this novel approach.
Analysis of marine biotoxins in shellfish Dr Andrew Turner (CEFAS Weymouth Laboratory, Weymouth, UK)
Paralytic shellfish poisoning algal toxins have been reported to cause severe illness in humans at 5.6-2058 μg Saxitoxin (STX) equivalents/kg and have a maximum permitted level of 800 μg STX equivalents/kg in shellfish tissue. Testing until 2006 was by mouse bio-assay and is still the official reference method. An alternative, chromatographic, method is post-column oxidation liquid chromatography with fluorescence detection (LC-FLD). However, this technique needs two columns/systems to run each sample, has very short column lifetimes and is prone to matrix effects giving false positive and negative results. Another approach uses hydrophobic interaction chromatography MS/MS (HILIC-MS/MS), but has sensitivity problems, long run-times, matrix interference, and ion-source fragmentation implications. Current work at CEFAS is investigating these analytical problems. Comparison of Xevo TQ® to Waters Acquity® showed a 10-50 times increase in sensitivity of the Xevo TQ®, which enabled very low limits of detection/quantification for standards. Fast UPLC was ideal for fast cycle time. Fragmentation mitigation included using negative mode rather than positive ionisation for some transitions, and optimisation of the HILIC separation. Matrix effects could not be removed by simple dilution techniques. Carbon solid-phase extraction removed 90-100% Na, Mg, K and Ca, although some Na formate remained. With these improvements the method was fully validated for 12 species of interest and showed good equivalence with the LC-FLD methods.
Pollution by polyisobutenes: a sticky problem for seabirds Professor Steve Rowland (University of Plymouth, Plymouth, UK)
Polyisobutenes (PIBs) are a generic group of oligomeric to polymeric, liquid to solid hydrocarbons, which are used in numerous industrial applications, including viscosity modifiers, lubricating oil additives, cosmetics (e.g. as lip glosses) and in chewing gum. PIBs have been manufactured since the 1930s, but currently they are made by well-controlled reactions, which produce alkenes and a larger proportion of oligomers with terminal double bonds. Some of these so-called ‘highly reactive’ (HR) PIBs, are then converted to other derivatives for use as ashless detergent additives for lubricants. These multiple uses require global movement of PIBs by road, rail and ship; some HR-PIB manufacturers are based in Europe, USA and Korea. Several marine pollution events involving PIBs or PIB derivatives have occurred, presumably from shipping discharges. Indeed the extant classification of PIBs in 2013 allowed for small legal discharges from ships at sea. In January and April 2013, discharges of what was identified by chemical analysis in Plymouth University laboratories as HR-PIB and confirmed by a laboratory in Germany, apparently polluted seabirds in the English Channel. Thousands of birds died and a media storm resulted. The background, analytical data, possible origins and a discussion of the possible fate of the HR-PIB spills were presented. Techniques used included NMR, infrared (IR) and high temperature GC-MS. The findings helped wildlife agencies (e.g. RSPCA, RSPB) to achieve a change in the classification of PIBs.
LC tandem MS strategies for the analysis of contaminants of emerging concern in water, soil and sediment samples Professor Damià Barceló (CSIC, Barcelona, Spain)
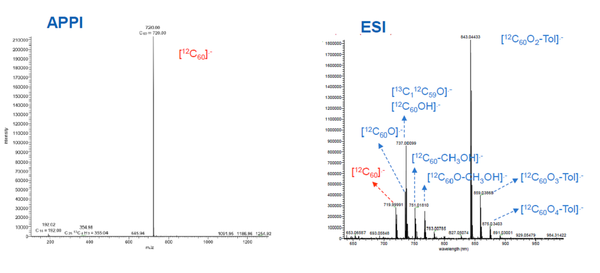
Endocrine disrupting compounds (EDCs) include a wide range of chemicals that are thought to disrupt the endocrine function of fish and mammals. Total EDC concentrations are found in the ng/L to μg/L range in river water samples and in the ng/g range in sediments. Concentrations were determined by a dual column LC switching system in which 2-5 mL sample were loaded onto a pre-concentration column, then eluted onto the analytical column with MS/MS analysis. On-line turbulent flow chromatography was used to investigate degradation products of tetracyclines after enzymatic degradation of contaminated waters. This was coupled to an LC-(ESI)-LTQ OrbitrapTM liquid chromatography-mass spectrometry system. Turbo flow uses large particle columns (30 μm or greater) and eliminates large molecules (e.g. peptides and proteins), and retains smaller molecules on the column. Separation is achieved by the difference in diffusion rates according to compound size. MS was achieved by full scan ESI, positive and negative ionisation, and ion trap with MS2 fragmentation of the five most intense peaks from the full scan. Iodinated X-ray contrast media compounds are frequently detected in wastewaters at μg/L concentrations and in surface waters up to 0.1 μg/L. The small number of commercially available standard compounds can readily be photolysed to produce over 100 transformation products. These were studied in laboratory samples, after simulated environmental degradation, and then in surface water samples. Once detected and prioritised, a number were isolated by semi-prep LC and their structures confirmed by 13C and 1H NMR spectroscopy. Initial screening was by high resolution MS, but once confirmed and a standard isolated, low resolution MS could be used. Fullerenes are emerging new pollutants and can have non-anthropogenic origins, such as volcanoes and forest fires, as well as industrial combustion-related sources. After extraction and chromatographic separation, fullerenes were analysed using atmospheric pressure photo-ionization MS (MS-APPI). This technique was found to be approximately 100 times more sensitive than MS-ESI (Figure 3).
Paralytic shellfish poisoning algal toxins have been reported to cause severe illness in humans at 5.6-2058 μg Saxitoxin (STX) equivalents/kg and have a maximum permitted level of 800 μg STX equivalents/kg in shellfish tissue. Testing until 2006 was by mouse bio-assay and is still the official reference method. An alternative, chromatographic, method is post-column oxidation liquid chromatography with fluorescence detection (LC-FLD). However, this technique needs two columns/systems to run each sample, has very short column lifetimes and is prone to matrix effects giving false positive and negative results. Another approach uses hydrophobic interaction chromatography MS/MS (HILIC-MS/MS), but has sensitivity problems, long run-times, matrix interference, and ion-source fragmentation implications. Current work at CEFAS is investigating these analytical problems. Comparison of Xevo TQ® to Waters Acquity® showed a 10-50 times increase in sensitivity of the Xevo TQ®, which enabled very low limits of detection/quantification for standards. Fast UPLC was ideal for fast cycle time. Fragmentation mitigation included using negative mode rather than positive ionisation for some transitions, and optimisation of the HILIC separation. Matrix effects could not be removed by simple dilution techniques. Carbon solid-phase extraction removed 90-100% Na, Mg, K and Ca, although some Na formate remained. With these improvements the method was fully validated for 12 species of interest and showed good equivalence with the LC-FLD methods.
Pollution by polyisobutenes: a sticky problem for seabirds Professor Steve Rowland (University of Plymouth, Plymouth, UK)
Polyisobutenes (PIBs) are a generic group of oligomeric to polymeric, liquid to solid hydrocarbons, which are used in numerous industrial applications, including viscosity modifiers, lubricating oil additives, cosmetics (e.g. as lip glosses) and in chewing gum. PIBs have been manufactured since the 1930s, but currently they are made by well-controlled reactions, which produce alkenes and a larger proportion of oligomers with terminal double bonds. Some of these so-called ‘highly reactive’ (HR) PIBs, are then converted to other derivatives for use as ashless detergent additives for lubricants. These multiple uses require global movement of PIBs by road, rail and ship; some HR-PIB manufacturers are based in Europe, USA and Korea. Several marine pollution events involving PIBs or PIB derivatives have occurred, presumably from shipping discharges. Indeed the extant classification of PIBs in 2013 allowed for small legal discharges from ships at sea. In January and April 2013, discharges of what was identified by chemical analysis in Plymouth University laboratories as HR-PIB and confirmed by a laboratory in Germany, apparently polluted seabirds in the English Channel. Thousands of birds died and a media storm resulted. The background, analytical data, possible origins and a discussion of the possible fate of the HR-PIB spills were presented. Techniques used included NMR, infrared (IR) and high temperature GC-MS. The findings helped wildlife agencies (e.g. RSPCA, RSPB) to achieve a change in the classification of PIBs.
LC tandem MS strategies for the analysis of contaminants of emerging concern in water, soil and sediment samples Professor Damià Barceló (CSIC, Barcelona, Spain)
Endocrine disrupting compounds (EDCs) include a wide range of chemicals that are thought to disrupt the endocrine function of fish and mammals. Total EDC concentrations are found in the ng/L to μg/L range in river water samples and in the ng/g range in sediments. Concentrations were determined by a dual column LC switching system in which 2-5 mL sample were loaded onto a pre-concentration column, then eluted onto the analytical column with MS/MS analysis. On-line turbulent flow chromatography was used to investigate degradation products of tetracyclines after enzymatic degradation of contaminated waters. This was coupled to an LC-(ESI)-LTQ OrbitrapTM liquid chromatography-mass spectrometry system. Turbo flow uses large particle columns (30 μm or greater) and eliminates large molecules (e.g. peptides and proteins), and retains smaller molecules on the column. Separation is achieved by the difference in diffusion rates according to compound size. MS was achieved by full scan ESI, positive and negative ionisation, and ion trap with MS2 fragmentation of the five most intense peaks from the full scan. Iodinated X-ray contrast media compounds are frequently detected in wastewaters at μg/L concentrations and in surface waters up to 0.1 μg/L. The small number of commercially available standard compounds can readily be photolysed to produce over 100 transformation products. These were studied in laboratory samples, after simulated environmental degradation, and then in surface water samples. Once detected and prioritised, a number were isolated by semi-prep LC and their structures confirmed by 13C and 1H NMR spectroscopy. Initial screening was by high resolution MS, but once confirmed and a standard isolated, low resolution MS could be used. Fullerenes are emerging new pollutants and can have non-anthropogenic origins, such as volcanoes and forest fires, as well as industrial combustion-related sources. After extraction and chromatographic separation, fullerenes were analysed using atmospheric pressure photo-ionization MS (MS-APPI). This technique was found to be approximately 100 times more sensitive than MS-ESI (Figure 3).
Fullerenes were extracted from water and sediments by liquid-liquid extraction and ultrasonic extraction, respectively. A pyrenylpropyl-bonded silica chromatographic column (buckyprep column) was used, with toluene as the mobile phase. Concentrations of C60 and C70 were found in the high pg-μg/L range in wastewater effluents, and concentrations were approximately a factor of 10 lower in river water close to the effluents. There was no clear relationship between the river water and discharge concentrations, which would suggest other inputs (e.g. atmospheric deposition). C60 and C70 were detected in 91% of urban soils samples studied in low pg/g concentrations.
Aspects of drinking water quality: the analysis of disinfection by-products Gavin Mills (Severn Trent Water Ltd., UK)
Analysis of halogenated and other compounds produced by disinfection is required in order to comply with drinking water supply quality standards. Trihalomethanes (THMs) have a PCV (prescribed concentration or value) of 1000 μg/L. De-chlorinated water samples are analysed by headspace GC-MS, with sodium chloride added as a matrix modifier. On-line monitoring can reduce costs by minimisation of additional treatment processes. Bromate, produced by ozone treatment of raw water containing bromide (PCV = 10 μg/L) can be analysed by ion-exchange chromatography detected by post-column derivatisation and UV, bromide, ClO2– and ClO3– detectable by conductivity. N-Nitrosodimethylamine is produced by ozonolysis of water containing specific pesticides (Action Levels 1, 10, 200 ng/L). Analysis is prone to interference if simple GC-MS is used. Confirmation can be made by GC-triple quadrupole MS (GC-QQQ). Analysis of halophenols by GC-QQQ allows reduced sample volumes. and automated sample preparation with the use of an Instrument Top Sample Prep cartridge.
Screening for novel anti-androgenic contaminants in environmental samples using mass spectro-metry combined with bio-assay procedures. Professor Elizabeth Hill (University of Sussex, Sussex, UK)
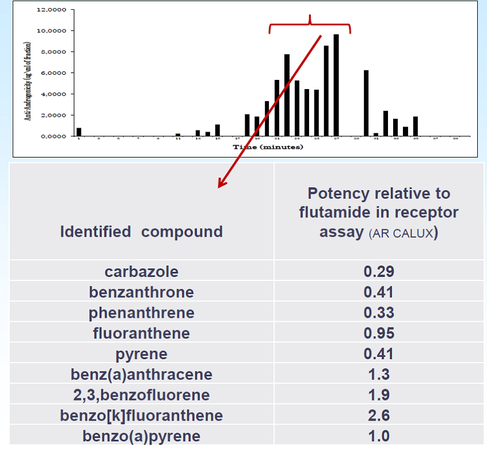
Although the concentrations of anti-androgenic compounds present in environmental samples are too low to have a direct effect on fish, they have previously been shown to contribute to feminisation of fish when in combined with estrogens. Professor Hill described a screening process for detecting anti-androgenic compounds using GC-MS and liquid chromatography/ time-of-flight MS (LC-TOFMS) combined with a bio-assay. Samples downstream of sewage effluent were extracted by silicone strips or low-density polyethylene flat tubing for compounds with log KOW > 4. For compounds with log KOW < 4, Polar Organic Chemical Integrative Samplers with Oasis HLB sorbent were used for pharmaceuticals and ENV+/Ambersorb/Biobeads for pesticides. The extracts were fractionated by reversed-phase HPLC, and fractions tested for anti-androgenic activity. Further studies identified anti-androgens in clams exposed to waste-water effluent and widespread anti-androgenic activity in coastal sediments around the English Channel (Figure 4). Bio-assay-directed analyses can be a critical tool to identify causative agents for toxicity.
Analysis of halogenated and other compounds produced by disinfection is required in order to comply with drinking water supply quality standards. Trihalomethanes (THMs) have a PCV (prescribed concentration or value) of 1000 μg/L. De-chlorinated water samples are analysed by headspace GC-MS, with sodium chloride added as a matrix modifier. On-line monitoring can reduce costs by minimisation of additional treatment processes. Bromate, produced by ozone treatment of raw water containing bromide (PCV = 10 μg/L) can be analysed by ion-exchange chromatography detected by post-column derivatisation and UV, bromide, ClO2– and ClO3– detectable by conductivity. N-Nitrosodimethylamine is produced by ozonolysis of water containing specific pesticides (Action Levels 1, 10, 200 ng/L). Analysis is prone to interference if simple GC-MS is used. Confirmation can be made by GC-triple quadrupole MS (GC-QQQ). Analysis of halophenols by GC-QQQ allows reduced sample volumes. and automated sample preparation with the use of an Instrument Top Sample Prep cartridge.
Screening for novel anti-androgenic contaminants in environmental samples using mass spectro-metry combined with bio-assay procedures. Professor Elizabeth Hill (University of Sussex, Sussex, UK)
Although the concentrations of anti-androgenic compounds present in environmental samples are too low to have a direct effect on fish, they have previously been shown to contribute to feminisation of fish when in combined with estrogens. Professor Hill described a screening process for detecting anti-androgenic compounds using GC-MS and liquid chromatography/ time-of-flight MS (LC-TOFMS) combined with a bio-assay. Samples downstream of sewage effluent were extracted by silicone strips or low-density polyethylene flat tubing for compounds with log KOW > 4. For compounds with log KOW < 4, Polar Organic Chemical Integrative Samplers with Oasis HLB sorbent were used for pharmaceuticals and ENV+/Ambersorb/Biobeads for pesticides. The extracts were fractionated by reversed-phase HPLC, and fractions tested for anti-androgenic activity. Further studies identified anti-androgens in clams exposed to waste-water effluent and widespread anti-androgenic activity in coastal sediments around the English Channel (Figure 4). Bio-assay-directed analyses can be a critical tool to identify causative agents for toxicity.
New developments in GC/MS and GC/PID instrumentation for rapid on-site analysis of trace environmental contaminants Dr Andrew Hobson (Quantitech Ltd, Milton Keynes, UK)
Commercial GC-photoionisation and GC-toroidal ion trap instruments have a range of environmental applications in the field. The GC-toroidal ion trap instrument (Figure 5) incorporates a capillary column, heating wire and temperature sensor wrapped together.
Commercial GC-photoionisation and GC-toroidal ion trap instruments have a range of environmental applications in the field. The GC-toroidal ion trap instrument (Figure 5) incorporates a capillary column, heating wire and temperature sensor wrapped together.
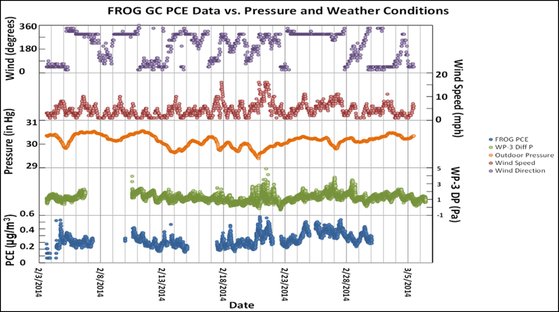
Typically, samples are introduced after pre-concentration by solid-phase microextraction. Examples of use include halomethanes in water over the range 10-100 ppb and geosmin (octahydro-4,8a-dimethyl-4a(2H)-naphthalenol) (responsible for odour in rain and earthy taste in fish) at 1ppt in water. The GC photoionisation instrument incorporates a pre-concentration stage where compounds sparged from a sample are absorbed on an application-specific sol gel. Heating releases the concentrate onto the 2.5 m, 0.8 μm OV-1 GC column. A typical application would be the analysis of ten volatile organic compounds in six minutes using a 40-100 ℃ temperature gradient. The portable nature of the instruments means that they can be left in situ for long-term analysis of contaminants. Figure 6 shows the influence of metrological conditions on tetrachloroethylene intrusion into an indoor environment from contaminated sub-soil.
The meeting closed with general agreement of a thoroughly worthwhile day introducing many new methods for complex matrices using minimal sample clean-up. A similar meeting is likely to be held in early 2017.
These abstracts were prepared by Roger Reeve, Graham Mills and Ian Forber based on the presentations and slides supplied by the speakers. The industry meeting sponsors were Agilent Technologies, Crawford Scientific, LGC, Markes International, SCIEX, Shimadzu, Sigma-Aldrich, Thames Restek, Thermo Scientific and Waters. Financial support is also acknowledged from ESED.
Presentation slides are available from www.rsc.org/Membership/Networking/InterestGroups/Environmental/PastEvents
These abstracts were prepared by Roger Reeve, Graham Mills and Ian Forber based on the presentations and slides supplied by the speakers. The industry meeting sponsors were Agilent Technologies, Crawford Scientific, LGC, Markes International, SCIEX, Shimadzu, Sigma-Aldrich, Thames Restek, Thermo Scientific and Waters. Financial support is also acknowledged from ESED.
Presentation slides are available from www.rsc.org/Membership/Networking/InterestGroups/Environmental/PastEvents