Mercury: history, facts and concerns
Exeter University
ECG Bulletin January 2022
Mercury is a unique and fascinating metallic element. Unlike other metals, it is a poor conductor, has a low melting point, and is liquid at standard temperature and pressure. This is due to its peculiar electronic configuration; electrons fill up as far as the 6s shell and are strongly influenced by relativistic atomic effects (1). The two 6s electrons move fast, and are tightly bound to the nucleus, increasing their relative mass, such that they do not contribute significantly to metal-metal bonds. The resulting weak bonding and reluctance to exchange electrons give mercury hints of noble gas behaviour, the best explanation for its liquid state. Mercury also has a high density, and metals like lead or copper float on it, while its high vapour pressure means that it slowly evaporates upon exposure to air. Mercury also has the highest surface tension of all liquids, which inhibits capillary action applied by absorbent materials, making it difficult to clean up spills.
Perhaps the most striking feature of mercury is its extreme toxicity. When it enters a living organism, it can substitute for cofactors in enzymatic reactions, interfering with biochemical transformations and metabolism (2). Protein function and ATP production are affected, and cellular activity eventually becomes damaged. This disruption can occur in cells from different organs, but it particularly affects the brain neurons (3). Consequently, people suffering from mercury poisoning typically suffer neurological symptoms such as speech difficulties, lack of coordination, vision loss, sensory conflicts, and tremors, among many others. Prolonged exposure to high concentrations of mercury compounds can ultimately lead to death. While all forms of mercury are toxic, organo-mercury compounds such as methylmercury (MeHg+, CH3Hg+) are particularly problematic because they accumulate through the food chain, causing concentrations to magnify in higher organisms.
Mercury is a scarce element in the Earth's crust, yet there is evidence of widespread use in ancient cultures. It is most commonly found as cinnabar (mercury sulphide, HgS), a bright red mineral and quintessential ore whose oldest known use is as a pigment. Cinnabar pigments have been found in pictograms of Egyptian tombs, American amulets and ceramics, Greek ointments, Roman cosmetics, and Chinese burials. It is a historical paradox that such a toxic element is used in such a diverse array of applications in medicine. From the 17th to 19th century, mercurous chloride was a popular syphilis treatment in Europe. Other mercury compounds served as dermal antiseptics to treat injuries. Mercury-containing amalgams became prevalent for dental fillings, and the preservative thiomersal (an organomercury derivative) has been used in vaccine manufacturing. Its diverse properties also made mercury popular for many other uses including batteries, switches, thermometers, manometers, light bulbs, household products, fungicides, and even explosives.
European alchemists thought mercury transcended physical boundaries, that it was the main ingredient in all metals, and could undergo transmutation into gold (4). Despite this belief, the ancients did discover mercury's amalgamation properties, setting the foundations for its most application today – in artisanal gold mining.
The use of mercury amalgamation to extract precious metals dates back to the Romans, around 50 AD (5). Amalgamation is the formation of a mercury alloy with another metal, such as gold. This preferential bonding extracts the precious metal from the ore by generating an amalgam that can be manually separated and burned to isolate the gold – the principal procedure utilised in artisanal and small-scale gold mining (Figure 1). This single activity introduced 1220 tonnes of mercury into terrestrial and freshwater environments in 2015 (6).
Although mercury's environmental and health effects are now well known, artisanal miners have practised amalgamation for centuries, making it an entrenched tradition that is hard to change. Mercury toxicity has been contested because poisoning becomes evident only after long-term exposure, and this viewpoint interferes with efforts to introduce alternative gold mining techniques, e.g. borax extraction (7). Consequently, to overcome mercury amalgamation's extensive use, it is necessary to use societal approaches to encourage the appropriation of safer procedures among ancestral mining communities
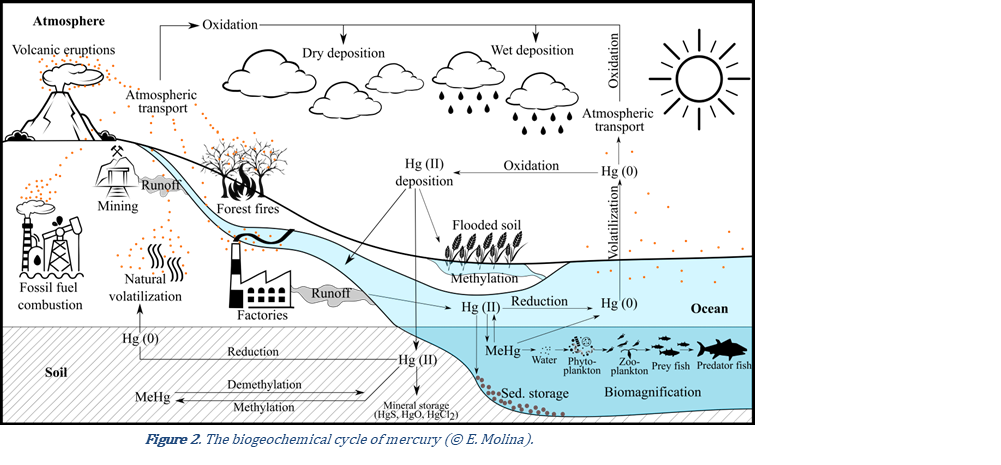
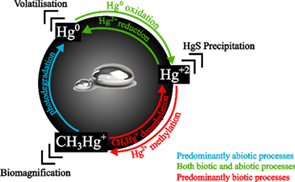
Mercury exists in three oxidation states: Hg0 (metallic), Hg+ (mercurous) and Hg2+ (mercuric, ionic) mercury. The latter forms a variety of inorganic as well as organometallic compounds (8). The speciation of Hg affects its behaviour in the environment and its toxicity. Metallic mercury vapour is emitted to the atmosphere by natural degassing of the Earth's surface, forest fires, or volcanic activity (Figure 2). However, most emissions and releases derive from anthropogenic sources, increasing total atmospheric concentrations to ~450% above natural levels (6). In the gaseous phase, mercury is transported by wind over long distances for up to a year before precipitating in aquatic and terrestrial ecosystems as oxidised Hg2+ (9). For example, in the polar regions, springtime mercury depletion events refer to low concentrations of gaseous elemental mercury due to its active oxidation and deposition onto ice and snow (10,11). Such favourable conditions for mercury accumulation pose high risks to humans and wildlife. After deposition, mercury may undergo transformation. In aerobic conditions, Hg0 is scarce in soils due to its high volatility and susceptibility to oxidise and, therefore, Hg2+ is the most common form (12). Hg2+ sorbs to soil constituents such as clay particles and organic matter. This process is influenced by several factors including the presence of chloride, dissolved organic carbon, and pH (13). After stabilisation, storage may last for decades (14). Microorganisms are able to survive in environments with high concentrations of toxic metals and can perform mercury speciation through their metabolism. Many prokaryotes have mer genes which confer resistance to mercury (15). Certain microorganisms can sequester, reduce, or oxidise mercury. Some microorganisms transform mercury between inorganic and organometallic compounds, producing methylmercury (Figure 2) and demethylating MeHg+ to produce Hg2+. Although many mercury transformation reactions can occur abiotically, methylation is strongly mediated by microbial activity. Genes associated with mercury methylation have been found across a range of redox conditions, including within sulphate-reducing bacteria found in anoxic conditions (16). In reduced sulfidic environments, considerable amounts of methylmercury become available for biomagnification up the trophic chain (15). This is significant in aquatic systems; biomagnification of MeHg+ in shellfish and fish was discovered to have caused a mass poisoning in Minamata, Japan in the 1950s. Bacteria can also reduce Hg2+ back to Hg0, leading to an increased probability of volatilisation (Figure 3).
|
Environmental challenges
Mercury pollution is a critical worldwide issue. Its toxic and non-degradable nature cause severe damage to air, water, and soil quality. The recent exclusion of mercury from most industrial applications is a step forward, yet many challenges lay ahead. The last UN Global Mercury Assessment states that the majority of emissions in 2015 occurred in Asia (49%), South America (18%), and Sub-Saharan Africa (16%). Artisanal gold mining accounts for almost 38% of the total anthropogenic global mercury emissions, and fossil fuel combustion for 24% (primarily from coal-burning (21%), as well as other fossil fuels (3%)) (6). Implementing control measures for these two biggest contributors is the only effective solution to solving the issue of global mercury pollution. |
Further, humanity must address the massive levels of mercury pollution already present in the environment. Scientific research has provided many ideas for how mercury pollution can be remediated. Activated carbon membranes have been used to filter atmospheric mercury, along with similar barrier systems and nanomaterials for water treatment (17-20). Teng et al. provide an excellent review of physical, chemical, and biological processes for removing or immobilising mercury in soils (21):
- replacement of polluted soil for clean soil
- soil vapour extraction (via reducing the vapour pressure of soil pores)
- thermal desorption to vaporise and trap mercury by heating
- electrokinetic remediation by establishing an electric field gradient using electrodes
- soil washing to extract and separate contaminants by eluent
- chemical stabilisation using immobilising reagents
- phytoremediation
- microbial remediation for binding, immobilisation, oxidation, transformation, and volatilisation
- Norrby, L. J. (1991). Why is mercury liquid? Or, why do relativistic effects not get into chemistry textbooks? J. Chem. Educ. 68, 110.
- Broussard, L. A., et al. (2002). The toxicology of mercury. Lab. Med. 33, 614–625.
- Cariccio, V. L. et al. (2019). Mercury Involvement in neuronal damage and in neurodegenerative diseases. Biol .Trace Elem. Res. 187, 341–356.
- Blum, J. D. (2013). Mesmerized by mercury. Nature Chem. 5, 1066–1066.
- de Lacerda, L. D., Salomons, W. (1998). The use of mercury amalgamation in gold and silver mining, In: de Lacerda, L. D., Salomons, W. (Eds.), Mercury from Gold and Silver Mining: A Chemical Time Bomb? Springer, Berlin, pp. 1–13.
- UNEP (2019). Global Mercury Assessment 2018. United Nations Environment Programme, Chemicals and Health Branch Geneva, Switzerland.
- Appel, P. W. U., Na-Oy, L. (2012). The borax method of gold extraction for small-scale miners. J. Health and Pollution 2, 5–10.
- WHO Regional Office for Europe (2000). Chapter 6.9 Mercury, in: Air Quality Guidelines. Copenhagen, Denmark.
- O’Driscoll, N., Rencz, A., Lean, D. (2005). The biogeochemistry and fate of mercury in the environment. Metal ions in Biological Systems 43, 221–38.
- Barkay, T., Poulain, A. J. (2007). Mercury (micro)biogeochemistry in polar environments, FEMS Microbiology Ecology 59, 232– 241.
- Steffen, A. et al. (2008). A synthesis of atmospheric mercury depletion event chemistry in the atmosphere and snow. Atmos. Chem. Phys. 8, 1445–1482.
- O’Connor, D. et al. (2019). Mercury speciation, transformation, and transportation in soils, atmospheric flux, and implications for risk management. A critical review. Environment International 126, 747–761.
- Newsome, L., Falagán, C. (2021). The microbiology of metal mine waste: bioremediation applications and implications for planetary health. Geohealth 5.
- Durão Jr, W. et al. (2009). Speciation, distribution, and transport of mercury in contaminated soils from Descoberto, Minas Gerais, Brazil. J. Environmental Monitoring 11, 1056–63.
- Mason, R. P. (2015). Chapter Sixteen: Geomicrobiology of Mercury, in: Ehrlich, H.L., Newman, D.K., Kappler, A. (Eds.), Ehrlich’s Geomicrobiology. CRC Press, Boca Raton, FL, pp. 323–342.
- Lin, H. et al. (2021). Mercury methylation by metabolically versatile and cosmopolitan marine bacteria. ISME J 15, 1810–1825.
- Albatrni, H., Qiblawey, H., El-Naas, M. H. (2021). Comparative study between adsorption and membrane technologies for the removal of mercury. Separation and Purification Technology 257, 15.
- Kawsher, S. (2015). Removal of mercury from wastewater by nanoparticle pyrite and ultrafiltration membrane system. Texas A&M University.
- Kim, C.S., Rytuba, J. J., Jr, G.E.B, (2004). Geological and anthropogenic factors influencing mercury speciation in mine wastes: An EXAFS spectroscopy study. Applied Geochemistry 19, 379–393.
- Wang, L. et al. (2020). Remediation of mercury contaminated soil, water, and air: A review of emerging materials and innovative technologies. Environment International 134, 105281.
- Teng, D. et al. (2020). Describing the toxicity and sources and the remediation technologies for mercury-contaminated soil. RSC Adv. 10, 23221–23232.
- EPA (1999). Phytoremediation Resource Guide.
- Rodriguez, et al. (2021). Promoting Responsible Recovery and Handling of Mercury from Contaminated Artisanal Gold Mining Tailings in Colombia. Technical Report. Published by the United States of America Department of State.