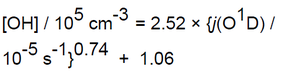Free radicals at high latitudes
Professor Dwayne Heard
School of Chemistry,
University of Leeds
ECG Bulletin January 2008
School of Chemistry,
University of Leeds
ECG Bulletin January 2008
Free radicals are chemical species which contain one or more unpaired electrons and thus are highly reactive. Free radicals are at the heart of the chemical cycles that process natural and anthropogenic emissions. Understanding atmospheric free radical chemistry, both in qualitative and quantitative terms, is essential for the construction of models of future changes to the atmosphere (and to the climate), as Prof. Dwayne Heard explains.
Introduction
Calculating the future change in the composition of our atmosphere is performed using complex climate change and air quality models. These models require the following information:
Emission control legislation, which globally is costing billions of dollars, is driven by model predictions so it is important that the inputs to the model are correct. We cannot wait to find out if the predictions are accurate. In order to probe whether the chemistry and photochemistry are faithfully represented by the model, we can use the chemical model to calculate key chemical intermediates that we can measure now.
Introduction
Calculating the future change in the composition of our atmosphere is performed using complex climate change and air quality models. These models require the following information:
- The future emissions into the atmosphere from anthropogenic and biogenic sources. A precise knowledge of such emissions is, of course, unknown, and assumptions have to be made using various emissions scenarios.
- Meteorological parameters, which control the mixing of emissions into the background atmosphere and effect long-range transport, for example wind velocities and larger atmospheric circulations.
- The detailed chemistry that controls the oxidative removal and processing of emissions via atmospheric free radical chemistry and photochemistry.
Emission control legislation, which globally is costing billions of dollars, is driven by model predictions so it is important that the inputs to the model are correct. We cannot wait to find out if the predictions are accurate. In order to probe whether the chemistry and photochemistry are faithfully represented by the model, we can use the chemical model to calculate key chemical intermediates that we can measure now.
Free radical chemistry in the atmosphere
Free radicals are ideal target species for atmospheric modelling, as their lifetimes are short (a few seconds) and their concentrations are influenced by the local in situ chemistry, and not by transport.
Free radicals are ideal target species for atmospheric modelling, as their lifetimes are short (a few seconds) and their concentrations are influenced by the local in situ chemistry, and not by transport.
Hydroxyl radical chemistry
The hydroxyl radical (OH), often referred to as the “detergent” of the atmosphere, reacts with virtually all trace species, regulating their concentrations and atmospheric lifetimes. Examples of species controlled by OH include CO and benzene (toxic substances), CH4 and HCFCs (greenhouse gases), hydrocarbons (smog precursors), oxygenated VOCs (precursors to secondary organic aerosols) and SO2 and NO2 (precursors to acidification).
It is important to measure OH as its concentration defines the oxidising capacity or self-cleansing capacity of the atmosphere, particularly in the daytime, and any trends are critical. OH is intimately involved in all aspects of the chemistry related to climate change and air pollution – and hence is an ideal target molecule for modelling. If a model can predict the concentration of OH successfully, we then have confidence that the chemistry is correct.
The hydroxyl radical (OH), often referred to as the “detergent” of the atmosphere, reacts with virtually all trace species, regulating their concentrations and atmospheric lifetimes. Examples of species controlled by OH include CO and benzene (toxic substances), CH4 and HCFCs (greenhouse gases), hydrocarbons (smog precursors), oxygenated VOCs (precursors to secondary organic aerosols) and SO2 and NO2 (precursors to acidification).
It is important to measure OH as its concentration defines the oxidising capacity or self-cleansing capacity of the atmosphere, particularly in the daytime, and any trends are critical. OH is intimately involved in all aspects of the chemistry related to climate change and air pollution – and hence is an ideal target molecule for modelling. If a model can predict the concentration of OH successfully, we then have confidence that the chemistry is correct.
Free radical chemistry in polar regions
Free radicals are generated by solar photolysis and until recently it was thought that areas of low insolation, such as the polar regions, were chemically rather barren. Models indicate that for the greenhouse gas methane, hardly any oxidation by reaction with OH occurs in the polar regions. However, recent fieldwork in polar regions has revealed elevated levels of free radicals (e.g. OH, HO2, RO2, IO and BrO), demonstrating the potential for significant chemical processing.
Free radicals are generated by solar photolysis and until recently it was thought that areas of low insolation, such as the polar regions, were chemically rather barren. Models indicate that for the greenhouse gas methane, hardly any oxidation by reaction with OH occurs in the polar regions. However, recent fieldwork in polar regions has revealed elevated levels of free radicals (e.g. OH, HO2, RO2, IO and BrO), demonstrating the potential for significant chemical processing.
|
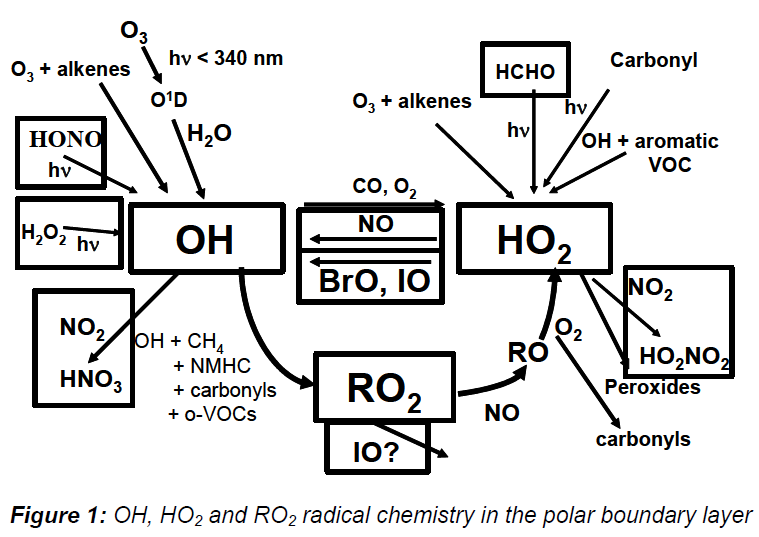
IO and BrO radicals are generated from reactions of I and Br atoms with ozone. The halogen atoms arise from the photolysis of photolabile halogens emitted from sea-weeds and other marine organisms. IO and BrO radicals can perturb the NO2/NO and HO2/OH ratios, and hence the oxidizing capacity of the atmosphere, as well as being involved in ozone depletion episodes and the oxidation of elemental mercury. Figure 1 summarizes the atmospheric chemistry of OH radicals.
Measurements of atmospheric free radicals The measurement of atmospheric free radicals is a difficult experimental challenge. The concentration of OH at noon is ~ 106 molecule cm-3 (mixing ratio of 1 part in 1014) and the lifetime is < 1 second. For other radicals, RO2, IO, BrO the concentrations are slightly larger at ~ 108 molecule cm-3, or a few parts per trillion. |
Techniques developed for their detection must be very sensitive, highly selective, and have good spatial and temporal resolution. In polar regions these radicals have been detected using laser-induced fluorescence (LIF; OH, HO2 after conversion to OH and IO), long-path differential optical absorption spectroscopy (DOAS; BrO, IO) and chemical ionization (CI) mass spectrometry (HO2 + RO2, OH).
Here at Leeds University both the LIF and DOAS methods have been deployed in polar regions, and the reaction sequence used to describe OH radicals is the Master Chemical Mechanism.
Hydroxyl radicals in the polar regions
Measurement of hydroxyl radicals. The first measurements of OH radicals in the polar regions were made at Palmer station on the Antarctic peninsula, during the SCATE campaign. Because the hydroxyl radicals were removed from local sources of halogen species, the chemistry behaved as expected for a remote pristine environment. The OH concentrations were very low (mean 24 hour [OH] = 1.1 x 105 molecule cm-3, solar noon [OH] ~ 5 x 105 molecule cm-3), and there was good agreement (30%) with a model calculation, showing that OH production was dominated by ozone photolysis followed by the reaction O(1D) + H2O, and its loss by reaction with CO and CH4. These measurements can be taken as a “base case”.
Measurement of hydroxyl radicals. The first measurements of OH radicals in the polar regions were made at Palmer station on the Antarctic peninsula, during the SCATE campaign. Because the hydroxyl radicals were removed from local sources of halogen species, the chemistry behaved as expected for a remote pristine environment. The OH concentrations were very low (mean 24 hour [OH] = 1.1 x 105 molecule cm-3, solar noon [OH] ~ 5 x 105 molecule cm-3), and there was good agreement (30%) with a model calculation, showing that OH production was dominated by ozone photolysis followed by the reaction O(1D) + H2O, and its loss by reaction with CO and CH4. These measurements can be taken as a “base case”.
Hydroxyl radical chemistry influences measured OH concentrations. Measurements of OH and HO2 + RO2 radicals taken at Summit Greenland (3207 m on the central plateau) showed a perturbation of the chemistry due to snow emissions of NOx and other oxidant precursors, for example formaldehyde (HCHO), hydrogen peroxide (H2O2) and nitrous acid (HONO), which are expected to augment levels of OH and HO2.
One mechanism for the production of HONO and NOx from snow is the photolysis of nitrate ions in the surface layer of snow. Difficulties were encountered in reproducing the measured levels of free radicals, partly due to uncertainties in the concentrations of key species, for example HONO, and unmeasured sources and sinks. Higher levels of OH were observed when air masses were being rapidly transported from coastal Greenland, providing some evidence for a possible influence of halogen chemistry.
One mechanism for the production of HONO and NOx from snow is the photolysis of nitrate ions in the surface layer of snow. Difficulties were encountered in reproducing the measured levels of free radicals, partly due to uncertainties in the concentrations of key species, for example HONO, and unmeasured sources and sinks. Higher levels of OH were observed when air masses were being rapidly transported from coastal Greenland, providing some evidence for a possible influence of halogen chemistry.
Nitrogen oxides measurements at the South Pole
There have been several field campaigns at the US-operated South Pole base, in which concentrations of free radicals have been measured. The results were most surprising. Elevated levels of nitric oxide [NO] = 100-200 pptv were observed, much higher than expected for a pristine, remote environment, but caused by snowpack emissions into a very shallow boundary layer (only a few 10s of m).
The higher NO concentration (Figure 1) can accelerate the catalytic cycles which regenerate OH, leading to elevated OH up to 1-2 x 106 molecule cm-3. As there is 24 hour sunlight at the South Pole, the oxidizing capacity in this shallow layer over much of Antarctica is higher than in the tropics! As for Summit Greenland, emissions of HCHO and H2O2 also contribute to enhanced photochemical activity, accounting ~ 25-30% each towards the production of OH and HO2.
Novel measurements of nitric acid (HNO3) and pernitric acid (HNO4) showed these species play an important role in the redeposition of nitrogen to the snowpack. In addition, because of the elevated NO, the rates of reactions HO2 + NO and RO2 + NO producing NO2 were fast, which generates in situ ozone following photolysis of ozone. Recent aircraft and balloon measurements from South Pole indicated that the enhanced production of ozone is quite widespread in the boundary layer above the Antarctic plateau. Despite a comprehensive set of measurements, models typically overpredicted OH concentrations, suggesting some misunderstandings of the chemistry still remain.
There have been several field campaigns at the US-operated South Pole base, in which concentrations of free radicals have been measured. The results were most surprising. Elevated levels of nitric oxide [NO] = 100-200 pptv were observed, much higher than expected for a pristine, remote environment, but caused by snowpack emissions into a very shallow boundary layer (only a few 10s of m).
The higher NO concentration (Figure 1) can accelerate the catalytic cycles which regenerate OH, leading to elevated OH up to 1-2 x 106 molecule cm-3. As there is 24 hour sunlight at the South Pole, the oxidizing capacity in this shallow layer over much of Antarctica is higher than in the tropics! As for Summit Greenland, emissions of HCHO and H2O2 also contribute to enhanced photochemical activity, accounting ~ 25-30% each towards the production of OH and HO2.
Novel measurements of nitric acid (HNO3) and pernitric acid (HNO4) showed these species play an important role in the redeposition of nitrogen to the snowpack. In addition, because of the elevated NO, the rates of reactions HO2 + NO and RO2 + NO producing NO2 were fast, which generates in situ ozone following photolysis of ozone. Recent aircraft and balloon measurements from South Pole indicated that the enhanced production of ozone is quite widespread in the boundary layer above the Antarctic plateau. Despite a comprehensive set of measurements, models typically overpredicted OH concentrations, suggesting some misunderstandings of the chemistry still remain.
Measurements of hydroxyl and hydroperoxyl (HO2) radicals during the CHABLIS campaign
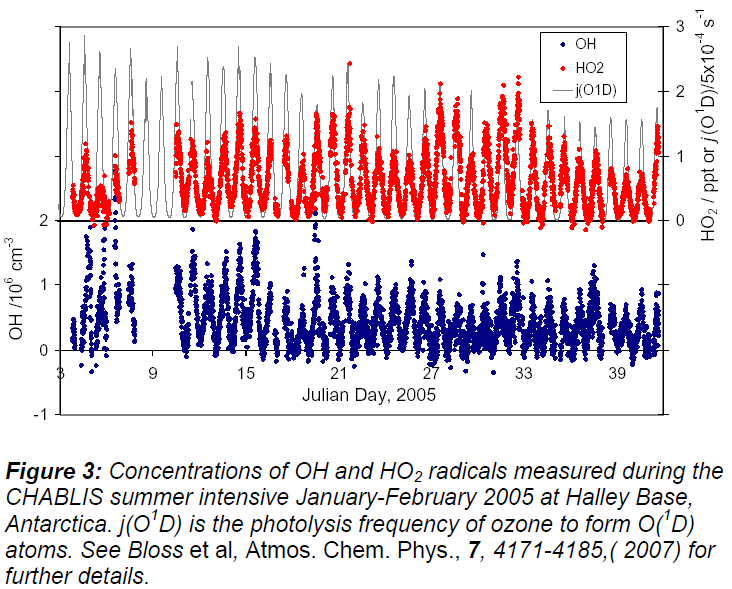
The CHABLIS (Chemistry of the Antarctic Boundary Layer and the Interface with Snow) campaign took place at the Halley Station, Antarctica. OH and HO2 radicals were measured in situ by LIF at low pressure (the FAGE technique) for a 2 month-intensive (January – February 2005), and BrO and IO radicals were measured year-round by DOAS, using an 8 km (2 x 4 km) light path ~4 - 5 m above the snowpack. Extensive coupling between these species was observed.
The CHABLIS (Chemistry of the Antarctic Boundary Layer and the Interface with Snow) campaign took place at the Halley Station, Antarctica. OH and HO2 radicals were measured in situ by LIF at low pressure (the FAGE technique) for a 2 month-intensive (January – February 2005), and BrO and IO radicals were measured year-round by DOAS, using an 8 km (2 x 4 km) light path ~4 - 5 m above the snowpack. Extensive coupling between these species was observed.
|
Halley is located on the floating Brunt Ice Shelf ~ 15 km from the ocean edge at 76 S, 26 W. The ice shelf is ~ 35 m thick and is moving at ~ 500 m per year. Local ice leads open up in the spring and summer months. The CHABLIS project was led by the British Antarctic Survey, but involved five UK Universities, and a wide range of trace gases and radiative parameters were measured. The FAGE instrument is housed in a shipping container and was positioned adjacent to the Clean Air Sector Laboratory (Figure 2). The container was unloaded from the supply ship onto the ice-shelf in December 2004, but by mid-late February 2005 the local ice conditions for that particular year made it impossible to load the container back onto the ship. Instead it was towed ~ 50 km over the ice to N9, an alternative loading site, and lifted back onto the ship, with undercut ice making the procedure rather precarious!
The depth of the mixed boundary layer is considerably larger at Halley than at South Pole, and so although there are still significant snow emissions, the concentration of NOx and HCHO is lower (mean NO = 8 ppt, NO2 = 5 ppt). Measurements of OH and HO2 taken over 37 days, with OH up to 2.5 x 106 molecule cm-3 observed early in the intensive period as shown in Figure 3. The relationship between [OH] and the photolysis frequency of ozone, j(O1D), was found to be:
with the less than linear relationship indicating considerable recycling of HO2 to OH mediated by reaction with NO, IO and BrO radicals (the latter both ~ 5 pptv in the summer). The non-zero OH when there is no photolysis of ozone indicates that OH is still generated by other processes, for example photolysis of HONO at considerably longer wavelengths.
|
The spring peak in BrO and IO (October) was ~ 20 pptv for both species, some of the highest levels ever recorded. These elevated levels are consistent with recent measurements of IO in this region from space. The IO and BrO are strongly correlated with local wind-direction and trajectory air-mass origins, and the highest concentrations were seen when air was sampled from the open ice front sector, where frost flowers are known to exist, providing one possible mechanism (but there are also others) for the production of short-lived halogen species.
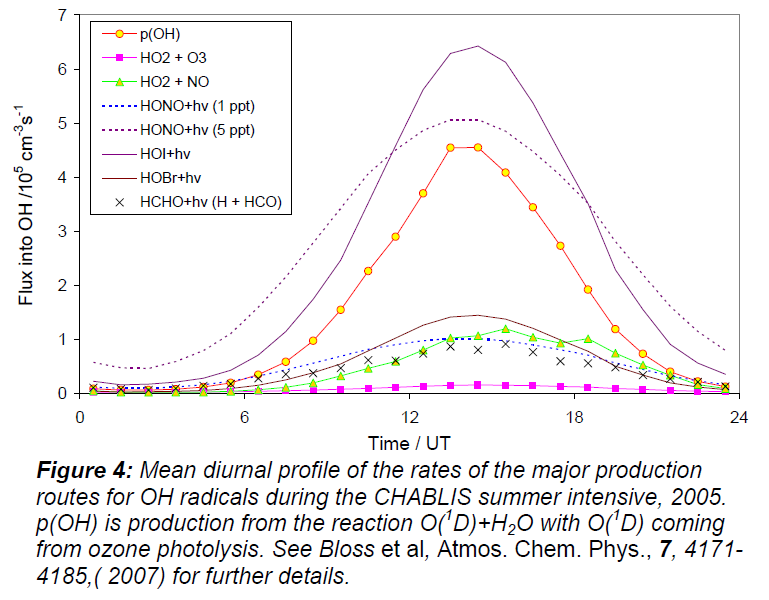
The main sources of HOx were the photolysis of O3 and HCHO, with important but uncertain contributions from HONO and higher aldehydes. CO and CH4 were the main sink for OH but model calculations suggest that other OH sinks are present, possibly oxygenated VOCs. The effect of BrO and IO on concentrations of OH and HO2 at Halley is quite striking. Figure 4 shows the calculated mean diurnal variation of the principal OH production mechanisms over the measurement period. The largest source of OH is the photolysis of HOI, with HOBr photolysis also important. HOI and HOBr are generated from the reactions HO2 + IO and HO2 + BrO, respectively. HONO is also an important but uncertain photolytic source of OH, and high quality HONO measurements are required.
|
Conclusion
There is now a considerable database of free radical measurements at high latitudes using a range of techniques. Free radicals are ideal targets to test the accuracy of chemical mechanisms. Oxidation chemistry mediated by OH is more complex than expected for a remote, pristine environment. Snow emissions of radical and NOx precursors increase the oxidising capacity of polar environments, and halogens control all important aspects of the chemistry of the coastal Antarctic boundary layer at Halley during the summer months. A key question is the vertical extent of halogen influence on the oxidative chemistry of polar regions. Recent satellite measurements suggest that elevated levels of IO exist throughout the boundary layer. Further fieldwork is planned in 2008 in the Arctic (Hudson Bay) to further investigate ozone depletion events, and the role of halogen species on the oxidative chemistry of the polar boundary layer. |
Acknowledgements
I would like to thank Dr. James Lee (York University) and Dr. William Bloss (University of Birmingham) who operated the Leeds FAGE instrument at Halley during the CHABLIS project, and other members of the CHABLIS consortium.
References
D. E. Heard and M. J. Pilling, “Measurement of OH and HO2 in the troposphere”, Chemical Reviews, 103, 5163-5198, (2003).
A. M. Grannas, A. E. Jones, J. Dibb, M. Ammann, C. Anastasio, H. J. Beine, M. Bergin, J. Bottenheim, C. S. Boxe, G. Carver, G. Chen, J. H. Crawford, F. Domin´e, M. M. Frey, M. I. Guzm´an, D. E. Heard, D. Helmig, M. R. Hoffmann, R. E. Honrath, L. G. Huey, M. Hutterli, H. W. Jacobi, P. Kl´an, B. Lefer, J. McConnell, J. Plane, R. Sander, J. Savarino, P. B. Shepson, W. R. Simpson, J. R. Sodeau, R. von Glasow, R. Weller, E. W. Wolff, and T. Zhu, “An overview of snow photochemistry: evidence, mechanisms and impacts”, Atmos. Chem. Phys. Discuss., 7, 4165–4283, (2007).
W. R. Simpson, R. von Glasow, K. Riedel, P. Anderson, P. Ariya, J. Bottenheim, J. Burrows, L. Carpenter, U. Frieß, M. E. Goodsite, D. Heard, M. Hutterli, H.-W. Jacobi, L. Kaleschke, B. Neff, J. Plane, U. Platt, A. Richter, H. Roscoe, R. Sander, P. Shepson, J. Sodeau, A. Steffen, T. Wagner, and E. Wolff, “Halogens and their role in polar boundary-layer ozone depletion”, Atmos. Chem. Phys. Discuss., 7, 4285–4403, (2007).
D. E. Heard (editor), Analytical techniques for atmospheric chemistry, Blackwell Publishing, 2006. ISBN 1405123575, 510 pp.
W. J. Bloss, J. D. Lee, D. E. Heard, R. A. Salmon, S. J.-B. Bauguitte, H. K. Roscoe, and A. E. Jones, “Observations of OH and HO2 radicals in coastal Antarctica”, Atmos. Chem. Phys. 7, 4171-4185, (2007).
A. Saiz-Lopez, J. M. C. Plane, A. S. Mahajan, P. S. Anderson, S. J.-B. Bauguitte, A. E. Jones, H. K. Roscoe, R. A. Salmon, W. J. Bloss, J. D. Lee, and D. E. Heard On the vertical distribution of boundary layer halogens over coastal Antarctica: implications for O3, HOx, NOx and the Hg lifetime Atmos. Chem. Phys. Discuss., 7, 9385-9417, (2007).
A. Saiz-Lopez, A. S. Mahajan, R. A. Salmon, S. J.-B. Bauguitte, A. E. Jones, H. K. Roscoe, and J.M. C. Plane, Boundary Layer Halogens in Coastal Antarctica, Science, 317, 348 – 351, (2007).
Professor DWAYNE HEARD
School of Chemistry,
University of Leeds,
Leeds LS2 9JT
This article is based on a presentation by Prof. Heard at the ECG’s 2007 Distinguished Guest Lecture and Symposium ‘Environmental Chemistry in the Polar Regions’. Other presentations from this meeting were published in the July 2007 issue of the ECG Bulletin.
Other accounts of the work at the Halley Station, Antarctica and the CHABLIS campaign appeared in issues of the ECG Bulletin published in July 2004, July 2006, and July 2007 respectively
I would like to thank Dr. James Lee (York University) and Dr. William Bloss (University of Birmingham) who operated the Leeds FAGE instrument at Halley during the CHABLIS project, and other members of the CHABLIS consortium.
References
D. E. Heard and M. J. Pilling, “Measurement of OH and HO2 in the troposphere”, Chemical Reviews, 103, 5163-5198, (2003).
A. M. Grannas, A. E. Jones, J. Dibb, M. Ammann, C. Anastasio, H. J. Beine, M. Bergin, J. Bottenheim, C. S. Boxe, G. Carver, G. Chen, J. H. Crawford, F. Domin´e, M. M. Frey, M. I. Guzm´an, D. E. Heard, D. Helmig, M. R. Hoffmann, R. E. Honrath, L. G. Huey, M. Hutterli, H. W. Jacobi, P. Kl´an, B. Lefer, J. McConnell, J. Plane, R. Sander, J. Savarino, P. B. Shepson, W. R. Simpson, J. R. Sodeau, R. von Glasow, R. Weller, E. W. Wolff, and T. Zhu, “An overview of snow photochemistry: evidence, mechanisms and impacts”, Atmos. Chem. Phys. Discuss., 7, 4165–4283, (2007).
W. R. Simpson, R. von Glasow, K. Riedel, P. Anderson, P. Ariya, J. Bottenheim, J. Burrows, L. Carpenter, U. Frieß, M. E. Goodsite, D. Heard, M. Hutterli, H.-W. Jacobi, L. Kaleschke, B. Neff, J. Plane, U. Platt, A. Richter, H. Roscoe, R. Sander, P. Shepson, J. Sodeau, A. Steffen, T. Wagner, and E. Wolff, “Halogens and their role in polar boundary-layer ozone depletion”, Atmos. Chem. Phys. Discuss., 7, 4285–4403, (2007).
D. E. Heard (editor), Analytical techniques for atmospheric chemistry, Blackwell Publishing, 2006. ISBN 1405123575, 510 pp.
W. J. Bloss, J. D. Lee, D. E. Heard, R. A. Salmon, S. J.-B. Bauguitte, H. K. Roscoe, and A. E. Jones, “Observations of OH and HO2 radicals in coastal Antarctica”, Atmos. Chem. Phys. 7, 4171-4185, (2007).
A. Saiz-Lopez, J. M. C. Plane, A. S. Mahajan, P. S. Anderson, S. J.-B. Bauguitte, A. E. Jones, H. K. Roscoe, R. A. Salmon, W. J. Bloss, J. D. Lee, and D. E. Heard On the vertical distribution of boundary layer halogens over coastal Antarctica: implications for O3, HOx, NOx and the Hg lifetime Atmos. Chem. Phys. Discuss., 7, 9385-9417, (2007).
A. Saiz-Lopez, A. S. Mahajan, R. A. Salmon, S. J.-B. Bauguitte, A. E. Jones, H. K. Roscoe, and J.M. C. Plane, Boundary Layer Halogens in Coastal Antarctica, Science, 317, 348 – 351, (2007).
Professor DWAYNE HEARD
School of Chemistry,
University of Leeds,
Leeds LS2 9JT
This article is based on a presentation by Prof. Heard at the ECG’s 2007 Distinguished Guest Lecture and Symposium ‘Environmental Chemistry in the Polar Regions’. Other presentations from this meeting were published in the July 2007 issue of the ECG Bulletin.
Other accounts of the work at the Halley Station, Antarctica and the CHABLIS campaign appeared in issues of the ECG Bulletin published in July 2004, July 2006, and July 2007 respectively