Criegee Intermediates: New atmospheric oxidants from old chemistry?
Atmospheric composition is affected by reaction with oxidant gases, mainly the hydroxyl radical (OH), the nitrate radical (NO3) and ozone (O3). These reactions initiate removal of reactive trace gases, including volatile organic compounds and some inorganic species such as sulphur dioxide (SO2). Until recently, only OH was thought to react directly with gas-phase SO2. This changed with the realisation that Criegee Intermediates (CIs) can react at rates relevant for atmospheric chemistry.
Atmospheric SO2 is the precursor to sulphate in aerosol particles. In addition to being harmful to human health (SO2 and sulphate aerosols were major components of the London “pea souper” smogs in the 1950s), sulphuric acid supports the formation of new particles, which affect climate by scattering solar radiation and affecting cloud droplet formation. Understanding SO2 processing is critical for accurate prediction of these effects on the atmosphere.
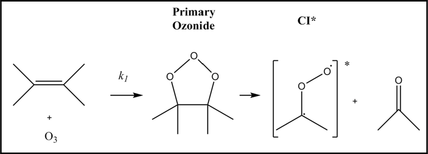
CIs (zwitterions or biradicals) form in the ozonolysis reaction between O3 and alkenes (Figure 1). They are named after Rudolf Criegee, who first proposed their existence in 1949 (1). In the gas phase, CIs can act as an additional atmospheric oxidant. Laboratory studies in the 1970s (2) showed that SO2 oxidation could be enhanced in the presence of alkenes and ozone, providing the first evidence that CIs could react with SO2. However, accepted wisdom was that reaction with water vapour dominates the removal of CIs in the atmosphere; CIs were thus not considered to be important oxidants.
Atmospheric SO2 is the precursor to sulphate in aerosol particles. In addition to being harmful to human health (SO2 and sulphate aerosols were major components of the London “pea souper” smogs in the 1950s), sulphuric acid supports the formation of new particles, which affect climate by scattering solar radiation and affecting cloud droplet formation. Understanding SO2 processing is critical for accurate prediction of these effects on the atmosphere.
CIs (zwitterions or biradicals) form in the ozonolysis reaction between O3 and alkenes (Figure 1). They are named after Rudolf Criegee, who first proposed their existence in 1949 (1). In the gas phase, CIs can act as an additional atmospheric oxidant. Laboratory studies in the 1970s (2) showed that SO2 oxidation could be enhanced in the presence of alkenes and ozone, providing the first evidence that CIs could react with SO2. However, accepted wisdom was that reaction with water vapour dominates the removal of CIs in the atmosphere; CIs were thus not considered to be important oxidants.
|
This picture changed following development of instrumental techniques capable of detecting CIs. Welz et al. used photoionisation mass spectrometry to make the first direct observations of individual CI isomers (3). They found that the reaction rates for the simplest CI, CH2OO, with SO2 were faster than expected by up to three orders of magnitude, whereas the CI + H2O reaction was comparatively slow. These results implied that CI reactions could contribute substantially to the atmospheric oxidation of SO2, increasing formation of sulphate aerosol particles. Measurements of sulphuric acid formation in forested regions (where alkene emissions are high) showed that additional oxidants for SO2, over and above OH, were present (4).
|
An explosion of research has followed, refining our understanding of the physical chemistry and atmospheric impact of CIs (5). Current understanding suggests that for the smallest CI, CH2OO, reaction with the water vapour dimer, (H2O)2, dominates atmospheric removal, with limited impacts on SO2, but larger CI species may contribute substantially to SO2 oxidation. A recent workshop (6) identified key priorities for future research as the need for further laboratory study of CI reaction rates, the development of new methods for synthesising CIs formed from larger alkenes, and the need for approaches to directly detect CIs in the atmosphere.
References
1. R. Criegee, G. Wenner, Liebigs Ann. Chem. 564, 9 (1949).
2. R. A. Cox, S. A. Penkett, Nature 230, 321 (1971).
3 O. Welz et al., Science 335, 204 (2012).
4 R. L. Mauldin III et al., Nature 488, 193 (2012).
5. C. A. Taatjes, D. E. Shallcross, C. J. Percival, Phys. Chem. Chem. Phys. 16, 1704 (2014).
6. Representation of Criegee Chemistry in Atmospheric Mechanisms, 29 February to 1 March 2016, Birmingham, UK. Supported by the ECG and the NERC [NE/K005448/1]
1. R. Criegee, G. Wenner, Liebigs Ann. Chem. 564, 9 (1949).
2. R. A. Cox, S. A. Penkett, Nature 230, 321 (1971).
3 O. Welz et al., Science 335, 204 (2012).
4 R. L. Mauldin III et al., Nature 488, 193 (2012).
5. C. A. Taatjes, D. E. Shallcross, C. J. Percival, Phys. Chem. Chem. Phys. 16, 1704 (2014).
6. Representation of Criegee Chemistry in Atmospheric Mechanisms, 29 February to 1 March 2016, Birmingham, UK. Supported by the ECG and the NERC [NE/K005448/1]