Veterinary antibiotics in the terrestrial environment
Melanie J. Bottoms
Department of Soil Science, University of Reading
ECB Bulletin July 2009
Department of Soil Science, University of Reading
ECB Bulletin July 2009
Although widely used, surprisingly little is known about the fate and chronic toxicity of some of the major classes of veterinary pharmaceuticals and their major metabolites in the terrestrial environment, as Melanie J. Bottoms, a final year PhD student from the Department of Soil Science, University of Reading explains.
Introduction
The last twenty years or so has witnessed increasing concern, both academic and public, regarding the fate of pharmaceuticals in the environment, and there is a growing body of knowledge regarding their presence and fate in numerous environmental matrices (Tolls, 2001; Boxall et al., 2002b). The majority of the work published to date has focussed heavily on aquatic systems, with an emphasis on investigating the fate of human pharmaceuticals in sewage treatment plants (Castiglioni et al., 2006; Göbel et al., 2004; Heberer, 2002) and drugs routinely used in aquaculture (Lai et al., 1995). Recently however, a number of workers have begun to consider the fate of veterinary antibiotics in the terrestrial environment.
A wide range of pharmaceuticals is used in the farming industry, for example sheep dips to protect animals from parasites; antibiotics for disease prevention and treatment; hormone treatments; growth promoters; vaccinations; and anthelmintics (wormers). It has been well documented that the chemicals utilised in sheep dips (organophosphates) are harmful to both human health and the environment (Boxall et al., 2004), and hormone treatments that have entered the environment can act as endocrine disrupters in fish (Nash et al., 2004). In 2003, it was reported that 100,000 to 200,000 tonnes of antibiotics alone were being used annually across the globe (Lindberg et al., 2004). It is clear that veterinary pharmaceuticals are widely used as both preventative medicine and treatment for a variety of animal ailments.
Routes of exposure
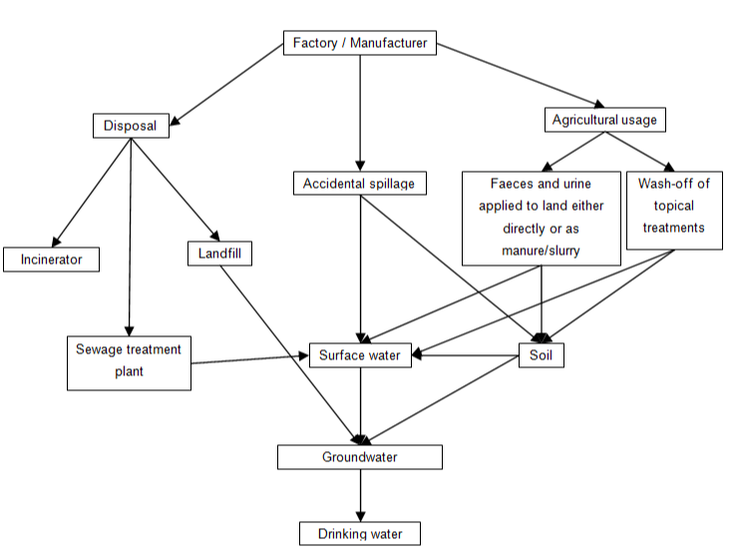
Figure 1 summarises the main routes of exposure to the environment for pharmaceuticals used in agriculture (excluding aquaculture). As can be seen, the primary routes of exposure are from the factory/manufacturer and from the end-user (in this case agriculture). It is unlikely that significant contamination will occur at the factory level, owing to the strict legislative controls to which pharmaceutical manufacturers are subjected.
Introduction
The last twenty years or so has witnessed increasing concern, both academic and public, regarding the fate of pharmaceuticals in the environment, and there is a growing body of knowledge regarding their presence and fate in numerous environmental matrices (Tolls, 2001; Boxall et al., 2002b). The majority of the work published to date has focussed heavily on aquatic systems, with an emphasis on investigating the fate of human pharmaceuticals in sewage treatment plants (Castiglioni et al., 2006; Göbel et al., 2004; Heberer, 2002) and drugs routinely used in aquaculture (Lai et al., 1995). Recently however, a number of workers have begun to consider the fate of veterinary antibiotics in the terrestrial environment.
A wide range of pharmaceuticals is used in the farming industry, for example sheep dips to protect animals from parasites; antibiotics for disease prevention and treatment; hormone treatments; growth promoters; vaccinations; and anthelmintics (wormers). It has been well documented that the chemicals utilised in sheep dips (organophosphates) are harmful to both human health and the environment (Boxall et al., 2004), and hormone treatments that have entered the environment can act as endocrine disrupters in fish (Nash et al., 2004). In 2003, it was reported that 100,000 to 200,000 tonnes of antibiotics alone were being used annually across the globe (Lindberg et al., 2004). It is clear that veterinary pharmaceuticals are widely used as both preventative medicine and treatment for a variety of animal ailments.
Routes of exposure
Figure 1 summarises the main routes of exposure to the environment for pharmaceuticals used in agriculture (excluding aquaculture). As can be seen, the primary routes of exposure are from the factory/manufacturer and from the end-user (in this case agriculture). It is unlikely that significant contamination will occur at the factory level, owing to the strict legislative controls to which pharmaceutical manufacturers are subjected.
The term “veterinary pharmaceutical” encompasses a wide variety of substances with a wide range of application methodologies. Each of these methods of application will result in a different route of exposure to the environment. Sheep-dip chemicals, for example, are applied topically and can be subject to washing-off by rainfall, resulting in contamination of surface waters and soil (Boxall et al., 2002b). Medications that are administered via injection, bolus or in feed, meanwhile, will be excreted by the animal in its urine or faeces, either as the parent compound or as a metabolite (Halling-Sørensen et al., 1998). These excretions are then stored as slurry, which is applied to the land as fertiliser, thus allowing veterinary pharmaceuticals to leach into groundwater or be transported in run-off to surface waters (Boxall et al., 2002a). Indeed, it has been estimated that approximately 75% of antimicrobials are excreted back to the environment (Campagnolo et al., 2002). Other routes of exposure include discharge (accidental or otherwise) from storage facilities, inappropriate disposal methods and input to river systems from aquaculture (Halling-Sørensen et al., 1998).
Analytical methods
There are no universally accepted analytical methodologies for the detection of veterinary pharmaceuticals in environmental matrices. Most methods utilise liquid chromatography coupled to a mass spectrometer. A few reviews have been published summarising the techniques that have been developed to analyse for human and veterinary pharmaceuticals in soils and sediments (e.g. Kim and Carlson, 2005 and Díaz-Cruz et al., 2003), and the most comprehensive overview is a summary by Boxall et al. (2002b).
Legal requirements
The use and manufacture of veterinary pharmaceuticals is highly regulated, to avoid both risk to human health and risk to the environment. It is now a legal requirement, under EU Directives 81/851/EEC and 81/852/EEC as codified by Directive 2001/82/EC and amended by 2004/28/EC, that an environmental risk assessment is carried out on any new veterinary medicine (Koschorreck et al., 2002), and on older medicines when their licences come up for renewal (Boxall et al., 2002b). This process requires acute toxicity tests to be conducted to determine the lethal concentration (LC50) or dose (LD50) of the drug on representative indicator species (e.g. microorganisms, plants, and earthworms for terrestrial ecosystems) (European Medicines Agency, 2004). Chronic tests are also required on earthworms. If there are then specific concerns, chronic toxicity tests may be conducted on other indicator species (European Medicines Agency, 2004). All of these test stages, however, are only carried out if there is an indication that the compound may pose an environmental risk (Boxall et al., 2002b) as determined by its Predicted Environmental Concentration (PEC) (European Medicines Agency, 2004). As such, these tests are not carried out routinely on all drugs. The tests are also limited in scope. It is unlikely that the pharmaceuticals will be present in such concentrations as to cause death, but organisms may be subject to chronic exposure, the sub-lethal effects of which are unknown (Svendsen et al., 2005). An additional problem with the use of acute toxicity tests was demonstrated by Wollenberger et al. (2000), who conducted standard acute toxicity tests, and OECD (Organisation for Economic Co-operation and Development) Reproduction Tests on Daphnia magna (a freshwater crustacean) using oxolinic acid, a pharmaceutical routinely used in aquaculture. They found that although the LC50 was established as 4.6 mg/l, no test organism survived the full three-week reproductive test at oxolinic acid concentrations greater than 0.75 mg/l, a concentration substantially smaller than that derived using data from the acute toxicity tests (Wollenberger et al., 2000).
Resistance
Veterinary pharmaceuticals in the environment pose not only a potential environmental threat, but also a very real threat to human health. Public awareness has been heightened to health risks following media publicity of so-called ‘super bugs’, like methicillin-resistant Staphylococcus aureus (MRSA), which have become resistant to antibiotics. Indeed, genes coding for resistance to one group of veterinary antibiotics, the tetracyclines, have been found in groundwater (Chee-Sanford et al., 2001), surface water (Knapp et al., 2008) and river sediments (Pruden et al., 2006). They are becoming so prevalent in environmental matrices that resistance genes are now referred to as emerging contaminants (Pruden et al., 2006). These genes are not just present in the environment, they are being taken up by organisms. For example, Ash et al. (2002) found organisms resistant to both naturally occurring and modified antibiotics in US rivers, a large proportion of which contained plasmids coding for resistance, and Engemann et al. (2008) found evidence that tetracycline resistance genes were being incorporated into water-column biofilms. Bacterial resistance can be developed by the insertion of foreign DNA into the chromosome from a plasmid (Smith, 2004), and these findings are particularly concerning. These concerns have been added to by work conducted by Knapp et al. (2008), who have demonstrated that anthropogenic tetracycline and oxytetracycline in surface waters can exert a resistance selection pressure. It is also believed that “contaminated” groundwater may act as a source of antibacterial resistant bacteria in the human-food chain (Campagnolo et al., 2002).
Veterinary antibiotics
A number of substances used in farming practice are known to be detrimental to the environment and have been well studied, for example sheep-dip chemicals (Boxall et al., 2002b) and anthelmintics (McCracken, 1993; Ridsdill-Smith, 1988; Strong, 1993; Wall and Strong, 1987; McKellar, 1997), but less is known about the fate, behaviour and effects of other groups of veterinary pharmaceuticals, e.g. antibiotics (Boxall et al., 2004). A number of drug families make up the class of pharmaceuticals commonly known as antibiotics. Two of the most widely used groups of antibiotics, tetracyclines and β-lactams, are discussed here.
Tetracyclines
The tetracyclines are possibly the most widely studied family of veterinary antibiotics. They are a broad-spectrum antibiotic, active against Mycoplasma, Chlamydophila and Rickettsia as well as a range of gram-positive and gram-negative bacteria (Russell, 2004). Of the tetracyclines, oxytetracycline is the most studied. It has been found to have an aerobic degradation half-life of 42-46 days, which was found to increase when sediment and sludge were added, and decrease when not in the presence of oxygen (Ingerslev et al., 2001). It has a low Kow (Boleas et al., 2005), a Koc of 27,792-93,317 (l/kg) (Rabølle and Spliid, 2000; Kay et al., 2004), and a Kd in sandy soil of 417 and in sandy loam soil of 1026 (Boleas et al., 2005). It is a mobile substance, which leaches readily and has been detected in rainfall runoff water (Kay et al., 2005c). However, pre-tilling soil prior to application of slurry has been found to reduce its movement through a macroporous clay soil (Kay et al., 2005a; Kay et al., 2005b). The application of manure can modify the fate and effects of oxytetracycline (Boleas et al., 2005). In fact, in an experiment by Boleas et al. in 2005, it was found that oxytetracycline (OTC) concentrations of 0.01 mg/kg soil promoted growth of wheat, causing a significant increase in biomass after 7 days in comparison to plants grown in control soil. However, when the oxytetracycline was administered with a co-addition of manure, inhibitory effects were observed at a concentration of 0.01 mg/kg soil. This suggests that the addition of manure increases toxicity to some plants (Boleas et al., 2005). OTC has been shown to reduce the growth of lettuce and carrot (grown in spiked soil) at environmentally realistic concentrations (Boxall et al., 2006). It inhibited shoot growth and root growth of alfalfa, by up to 61% and 85% respectively, for plants grown in a growth solution containing the drug at concentrations of 0.02 mM OTC (shoots) and >0.002 mM OTC (roots) (Kong et al., 2007). Kong et al. (2007) also demonstrated a linear uptake of OTC from a 0.004 mM OTC solution by alfalfa over 4 hours; uptake was both time and pH dependent.
In a series of bacterial tests measuring substrate-induced respiration in the presence of OTC with different soil types, it has been found that the more strongly sorbed the drug was the less bioavailable it was. Conversely, the less strongly sorbed it was the greater the inhibitory effect in the first instance, but the more quickly the bacterial community recovered (Thiele-Bruhn and Beck, 2005). The inhibitory effect of OTC on soil bacteria has also been demonstrated to be greater in the presence of copper, a common soil pollutant in both developing and developed industrial countries. Kong et al. (2006) found that both substances significantly decreased functional diversity and substrate use by bacteria. This effect was observed in the presence of just 11 μM oxytetracycline or 20 μM copper, but when the substances were applied together the effect was more pronounced, suggesting a synergistic effect (Kong et al., 2006).
β-Lactams
The β-lactam family is considered by some to be the most important group of antibiotics (Benito-Peña et al., 2006). β-Lactams include well-known groups of drugs, such as penicillins and cephalosporins (Russell, 2004). In 2004 it was reported that 50% of all antibiotics sold worldwide were β-lactams (Deshpande et al., 2004). Within the UK in 2007, the veterinary therapeutic antimicrobial market comprised 387 tonnes of antimicrobial active ingredient, nineteen percent of which were β-lactams (Goodyear, 2008). Of the products sold, thirty-five percent contained β-lactam drugs (Goodyear, 2008).
Most β-lactams are active against both gram-negative and gram-positive bacteria, (Cha et al., 2006; Pérez-Lozano et al., 2006). They are regularly used in dairy farming to treat mastitis in lactating cattle. Indeed, of 179 antibiotics utilised for mastitis treatment in Western Europe and the US, 75% are β-lactams (Riediker et al., 2004). Unfortunately, there is growing resistance to these drugs. In 2004 it was reported that 60% of Streptococcus pneumoniae strains were resistant to the most frequently used β-lactam antibiotics (Deshpande et al., 2004).
Amoxicillin is a high-use β-lactam. In 2007, 51 tonnes of active penicillin ingredient were sold to the UK veterinary market alone (this figure includes sales of amoxicillin, ampicillin, cloxacillin, nafcillin and panthamate hydroide) (Goodyear, 2008), whilst in Germany approximately 100 Mg of amoxicillin is prescribed annually (Schreiber and Szewzyk, 2008). Basic physico-chemical data are known for this compound and can be seen in Table 1. Like the other members of the β-lactam family, amoxicillin is readily degraded in the environment (Cha et al., 2006), but it has been detected in river water at concentrations less than 10 ng/l (Christian et al., 2003). Amoxicillin has also been detected in sewage-treatment plant effluents in Italy at concentrations up to 120 ng/l (Andreozzi et al., 2004).
Analytical methods
There are no universally accepted analytical methodologies for the detection of veterinary pharmaceuticals in environmental matrices. Most methods utilise liquid chromatography coupled to a mass spectrometer. A few reviews have been published summarising the techniques that have been developed to analyse for human and veterinary pharmaceuticals in soils and sediments (e.g. Kim and Carlson, 2005 and Díaz-Cruz et al., 2003), and the most comprehensive overview is a summary by Boxall et al. (2002b).
Legal requirements
The use and manufacture of veterinary pharmaceuticals is highly regulated, to avoid both risk to human health and risk to the environment. It is now a legal requirement, under EU Directives 81/851/EEC and 81/852/EEC as codified by Directive 2001/82/EC and amended by 2004/28/EC, that an environmental risk assessment is carried out on any new veterinary medicine (Koschorreck et al., 2002), and on older medicines when their licences come up for renewal (Boxall et al., 2002b). This process requires acute toxicity tests to be conducted to determine the lethal concentration (LC50) or dose (LD50) of the drug on representative indicator species (e.g. microorganisms, plants, and earthworms for terrestrial ecosystems) (European Medicines Agency, 2004). Chronic tests are also required on earthworms. If there are then specific concerns, chronic toxicity tests may be conducted on other indicator species (European Medicines Agency, 2004). All of these test stages, however, are only carried out if there is an indication that the compound may pose an environmental risk (Boxall et al., 2002b) as determined by its Predicted Environmental Concentration (PEC) (European Medicines Agency, 2004). As such, these tests are not carried out routinely on all drugs. The tests are also limited in scope. It is unlikely that the pharmaceuticals will be present in such concentrations as to cause death, but organisms may be subject to chronic exposure, the sub-lethal effects of which are unknown (Svendsen et al., 2005). An additional problem with the use of acute toxicity tests was demonstrated by Wollenberger et al. (2000), who conducted standard acute toxicity tests, and OECD (Organisation for Economic Co-operation and Development) Reproduction Tests on Daphnia magna (a freshwater crustacean) using oxolinic acid, a pharmaceutical routinely used in aquaculture. They found that although the LC50 was established as 4.6 mg/l, no test organism survived the full three-week reproductive test at oxolinic acid concentrations greater than 0.75 mg/l, a concentration substantially smaller than that derived using data from the acute toxicity tests (Wollenberger et al., 2000).
Resistance
Veterinary pharmaceuticals in the environment pose not only a potential environmental threat, but also a very real threat to human health. Public awareness has been heightened to health risks following media publicity of so-called ‘super bugs’, like methicillin-resistant Staphylococcus aureus (MRSA), which have become resistant to antibiotics. Indeed, genes coding for resistance to one group of veterinary antibiotics, the tetracyclines, have been found in groundwater (Chee-Sanford et al., 2001), surface water (Knapp et al., 2008) and river sediments (Pruden et al., 2006). They are becoming so prevalent in environmental matrices that resistance genes are now referred to as emerging contaminants (Pruden et al., 2006). These genes are not just present in the environment, they are being taken up by organisms. For example, Ash et al. (2002) found organisms resistant to both naturally occurring and modified antibiotics in US rivers, a large proportion of which contained plasmids coding for resistance, and Engemann et al. (2008) found evidence that tetracycline resistance genes were being incorporated into water-column biofilms. Bacterial resistance can be developed by the insertion of foreign DNA into the chromosome from a plasmid (Smith, 2004), and these findings are particularly concerning. These concerns have been added to by work conducted by Knapp et al. (2008), who have demonstrated that anthropogenic tetracycline and oxytetracycline in surface waters can exert a resistance selection pressure. It is also believed that “contaminated” groundwater may act as a source of antibacterial resistant bacteria in the human-food chain (Campagnolo et al., 2002).
Veterinary antibiotics
A number of substances used in farming practice are known to be detrimental to the environment and have been well studied, for example sheep-dip chemicals (Boxall et al., 2002b) and anthelmintics (McCracken, 1993; Ridsdill-Smith, 1988; Strong, 1993; Wall and Strong, 1987; McKellar, 1997), but less is known about the fate, behaviour and effects of other groups of veterinary pharmaceuticals, e.g. antibiotics (Boxall et al., 2004). A number of drug families make up the class of pharmaceuticals commonly known as antibiotics. Two of the most widely used groups of antibiotics, tetracyclines and β-lactams, are discussed here.
Tetracyclines
The tetracyclines are possibly the most widely studied family of veterinary antibiotics. They are a broad-spectrum antibiotic, active against Mycoplasma, Chlamydophila and Rickettsia as well as a range of gram-positive and gram-negative bacteria (Russell, 2004). Of the tetracyclines, oxytetracycline is the most studied. It has been found to have an aerobic degradation half-life of 42-46 days, which was found to increase when sediment and sludge were added, and decrease when not in the presence of oxygen (Ingerslev et al., 2001). It has a low Kow (Boleas et al., 2005), a Koc of 27,792-93,317 (l/kg) (Rabølle and Spliid, 2000; Kay et al., 2004), and a Kd in sandy soil of 417 and in sandy loam soil of 1026 (Boleas et al., 2005). It is a mobile substance, which leaches readily and has been detected in rainfall runoff water (Kay et al., 2005c). However, pre-tilling soil prior to application of slurry has been found to reduce its movement through a macroporous clay soil (Kay et al., 2005a; Kay et al., 2005b). The application of manure can modify the fate and effects of oxytetracycline (Boleas et al., 2005). In fact, in an experiment by Boleas et al. in 2005, it was found that oxytetracycline (OTC) concentrations of 0.01 mg/kg soil promoted growth of wheat, causing a significant increase in biomass after 7 days in comparison to plants grown in control soil. However, when the oxytetracycline was administered with a co-addition of manure, inhibitory effects were observed at a concentration of 0.01 mg/kg soil. This suggests that the addition of manure increases toxicity to some plants (Boleas et al., 2005). OTC has been shown to reduce the growth of lettuce and carrot (grown in spiked soil) at environmentally realistic concentrations (Boxall et al., 2006). It inhibited shoot growth and root growth of alfalfa, by up to 61% and 85% respectively, for plants grown in a growth solution containing the drug at concentrations of 0.02 mM OTC (shoots) and >0.002 mM OTC (roots) (Kong et al., 2007). Kong et al. (2007) also demonstrated a linear uptake of OTC from a 0.004 mM OTC solution by alfalfa over 4 hours; uptake was both time and pH dependent.
In a series of bacterial tests measuring substrate-induced respiration in the presence of OTC with different soil types, it has been found that the more strongly sorbed the drug was the less bioavailable it was. Conversely, the less strongly sorbed it was the greater the inhibitory effect in the first instance, but the more quickly the bacterial community recovered (Thiele-Bruhn and Beck, 2005). The inhibitory effect of OTC on soil bacteria has also been demonstrated to be greater in the presence of copper, a common soil pollutant in both developing and developed industrial countries. Kong et al. (2006) found that both substances significantly decreased functional diversity and substrate use by bacteria. This effect was observed in the presence of just 11 μM oxytetracycline or 20 μM copper, but when the substances were applied together the effect was more pronounced, suggesting a synergistic effect (Kong et al., 2006).
β-Lactams
The β-lactam family is considered by some to be the most important group of antibiotics (Benito-Peña et al., 2006). β-Lactams include well-known groups of drugs, such as penicillins and cephalosporins (Russell, 2004). In 2004 it was reported that 50% of all antibiotics sold worldwide were β-lactams (Deshpande et al., 2004). Within the UK in 2007, the veterinary therapeutic antimicrobial market comprised 387 tonnes of antimicrobial active ingredient, nineteen percent of which were β-lactams (Goodyear, 2008). Of the products sold, thirty-five percent contained β-lactam drugs (Goodyear, 2008).
Most β-lactams are active against both gram-negative and gram-positive bacteria, (Cha et al., 2006; Pérez-Lozano et al., 2006). They are regularly used in dairy farming to treat mastitis in lactating cattle. Indeed, of 179 antibiotics utilised for mastitis treatment in Western Europe and the US, 75% are β-lactams (Riediker et al., 2004). Unfortunately, there is growing resistance to these drugs. In 2004 it was reported that 60% of Streptococcus pneumoniae strains were resistant to the most frequently used β-lactam antibiotics (Deshpande et al., 2004).
Amoxicillin is a high-use β-lactam. In 2007, 51 tonnes of active penicillin ingredient were sold to the UK veterinary market alone (this figure includes sales of amoxicillin, ampicillin, cloxacillin, nafcillin and panthamate hydroide) (Goodyear, 2008), whilst in Germany approximately 100 Mg of amoxicillin is prescribed annually (Schreiber and Szewzyk, 2008). Basic physico-chemical data are known for this compound and can be seen in Table 1. Like the other members of the β-lactam family, amoxicillin is readily degraded in the environment (Cha et al., 2006), but it has been detected in river water at concentrations less than 10 ng/l (Christian et al., 2003). Amoxicillin has also been detected in sewage-treatment plant effluents in Italy at concentrations up to 120 ng/l (Andreozzi et al., 2004).
In aquatic environments, amoxicillin has been shown to have toxic effects on the blue-green alga Synechococcus leopolensis (EC50 = 2.22 μg/l) (Andreozzi et al., 2004), and inhibitory effects on biofilm formation of Aquabacterium commune (at 0.5 μg/l) and Escherichia coli (at 50 μg/l), whilst having positive effects for the formation of biofilm by E.coli (at both 0.5 μg/l and 50 μg/l) when also in the presence of iron (Schreiber and Szewzyk, 2008). In the soil environment very little work has been carried out. One study looked at the toxicity and uptake of amoxicillin by carrot and lettuce, no toxic effect or uptake being observed (Boxall et al., 2006), whilst another inconclusive study investigated whether the presence of amoxicillin in soil increased the proportion of amoxicillin resistant bacteria present in soil bacterial communities (Binh et al., 2007). Amoxicillin has received very little attention from researchers, especially when considered alongside the frequency of its use. This has been recognised within risk-assessment paradigms put forward in the literature, with both Boxall et al. (2003) and more recently Capleton et al. (2006) rating it as a high-priority substance for the acquisition of toxicity and fate data.
It is clear that further investigation of the environmental fate and effects of amoxicillin is required, but additionally the other β-lactams should not be overlooked. In the UK in 2007, only 6 tonnes[S1] of active antimicrobial ingredient of cephalosporins were sold, but this is increasing year on year and has nearly doubled since 2004 (3,240 kg in 2004 compared to 6,215 kg in 2007) (Goodyear, 2008). In the case of ceftiofur, a cephalosporin antibiotic, it is known that more than 95% of the dose is excreted within twenty-four hours of administration in the urine and faeces as a combination of the parent compound and the two major metabolites (desfuroylceftiofur and desfuroylceftiofur cysteine disulphide) (The European Agency for the Evaluation of Medicinal Products, 1999). The two major metabolites retain the β-lactam moiety (The European Agency for the Evaluation of Medicinal Products, 1999), and thus retain the potential for biological activity.
Conclusions
The environmental fate of veterinary products is still a developing area of research. Work needs to proceed rapidly in order to enable policy makers to act with the most robust scientific evidence to protect both human health, and our environmental legacy for future generations. In particular, research to determine the ecotoxicological effects of the β-lactam drugs on key terrestrial indicator species would help establish the high priority substances from an environmental perspective. Additionally, characterisation of the major degradation products of the high use veterinary antibiotics is required. This is especially the case for the β-lactams, which are prone to hydrolysis in the environment (Cha et al., 2006), as there is little knowledge regarding the properties of β-lactam degradation products.
Acknowledgements
The author would like to thank her funding bodies, the EPSRC and the University of Reading’s Research Endowment Trust Fund. She would also like to thanks her supervisors Dr. Mark E. Hodson, Dr. Andrew Parker and Prof. Rainer Cramer for their support.
References
Andreozzi, R.; Caprio, V.; Ciniglia, C.; De Champdoré, M.; Lo Giudice, R.; Marotta, R.; Zuccato, E. (2004), ‘Antibiotics in the environment: Occurrence in Italian STPs, fate, and preliminary assessment on algal toxicity of amoxicillin’, Environmental Science and Technology, 38, 6832-6838.
Ash, R. J.; Mauck, B.; Morgan, M. (2002), ‘Antibiotic resistance of gram-negative bacteria in rivers, United States’, Emerging Infectious Diseases, 8, 713-716.
Benito-Peña, E.; Partal-Rodera, A. I.; León-González, M. E.; Moreno-Bondi, M. C. (2006), ‘Evaluation of mixed mode solid phase extraction cartridges for the preconcentration of ß-lactam antibiotics in wastewater using liquid chromatography with UV-DAD detection’, Analytica Chimica Acta, 556, 415-422.
Binh, C. T. T.; Heuer, H.; Gomes, N. C. M.; Kotzerke, A.; Fulle, M.; Wilke, B.-M.; Schloter, M.; Smalla, K. (2007), ‘Short-term effects of amoxicillin on bacterial communities in manured soil’, FEMS Microbiology Ecology, 62, 290-302.
Boleas, S.; Alonso, C.; Pro, J.; Fernández, C.; Carbonell, G.; Tarazona, J. V. (2005), ‘Toxicity of the antimicrobial oxytetracycline to soil organisms in a multi-species-soil system (MS.3) and influence of manure co-addition’, Journal of Hazardous Materials, 122, 233-241.
Boxall, A. B. A.; Blackwell, P.; Cavallo, R.; Kay, P.; Tolls, J. (2002a), ‘The sorption and transport of a sulphonamide antibiotic in soil systems’, Toxicology Letters, 131, 19-28.
Boxall, A. B. A.; Fogg, L. A.; Blackwell, P. A.; Kay, P.; Pemberton, E. J. (2002b), ‘Review of veterinary medicines in the environment’, R&D Technical Report P6-012/8/TR, Cranfield Centre for EcoChemistry, Environment Agency, Bristol; available online http://publications.environment-agency.gov.uk/pdf/SP6-012-8-TR-e-p.pdf
Boxall, A. B. A.; Fogg, L. A.; Kay, P.; Blackwell, P. A.; Pemberton, E. J.; Croxford, A. (2003), ‘Prioritisation of veterinary medicines in the UK environment’, Toxicology Letters, 142, 207-218.
Boxall, A. B. A.; Fogg, L. A.; Kay, P.; Blackwell, P. A.; Pemberton, E. J.; Croxford, A. (2004), ‘Veterinary medicines in the environment’, Reviews of Environmental Contamination and Toxicology, 180, 1-91.
Boxall, A. B. A.; Johnson, P.; Smith, E. J.; Sinclair, C. J.; Stutt, E.; Levy, L. S. (2006), ‘Uptake of veterinary medicines from soils into plants’, Journal of Agricultural and Food Chemistry, 54, 2288-2297.
Campagnolo, E. R.; Johnson, K. R.; Karpati, A.; Rubin, C. S.; Kolpin, D. W.; Meyer, M. T.; Esteban, J. E.; Currier, R. W.; Smith, K.; Thu, K. M.; McGeehin, M. (2002), ‘Antimicrobial residues in animal waste and water resources proximal to large-scale swine and poultry feeding operations’, The Science of the Total Environment, 299, 89-95.
Capleton, A. C.; Courage, C.; Rumsby, P.; Holmes, P.; Stutt, E.; Boxall, A. B. A.; Levy, L.S. (2006), ‘Prioritising veterinary medicines according to their potential indirect human exposure and toxicity profile’, Toxicology Letters, 163, 213-223.
Castiglioni, S.; Bagnati, R.; Fanelli, R.; Pomati, F.; Calamari, D.; Zuccato, E. (2006), ‘Removal of pharmaceuticals in sewage treatment plants in Italy’, Environmental Science and Technology, 40, 357-363.
Cha, J. M.; Yang, S.; Carlson, K. H. (2006), ‘Trace determination of ß-lactam antibiotics in surface water and urban wastewater using liquid chromatography combined with electrospray tandem mass spectrometry’, Journal of Chromatography A, 1115, 46-57.
Chee-Sanford, J. C.; Aminov, R. I.; Krapac, I. J.; Garrigues-Jeanjean, N.; Mackie, R. I. (2001), ‘Occurrence and diversity of tetracycline resistance genes in lagoons and groundwater underlying two swine production facilities’, Applied and Environmental Microbiology, 67, 1494-1502.
Christian, T.; Schneider, R. J.; Färber, H. A.; Skutlarek, D.; Meyer, M. T.; Goldbach, H. E. (2003), ‘Determination of antibiotics residues in manure, soil, and surface waters’, Acta Hydrochimica et Hydrobiologica, 31, 34-44.
Deshpande, A. D.; Baheti, K. G.; Chatterjee, N. R. (2004), ‘Degradation of ß-lactam antibiotics’, Current Science, 87, 1684-1695.
Díaz-Cruz, M.; López de Alda, M. J.; Barceló, D. (2003), ‘Environmental behaviour and analysis of veterinary and human drugs in soils, sediments and sludge’, Trends in Analytical Chemistry, 22, 340-351.
Engemann, C. A.; Keen, P. L.; Knapp, C. W.; Hall, K. J.; Graham, D. W. (2008), ‘Fate of tetracycline resistance genes in aquatic systems: migration from the water column to peripheral biofilms’, Environmental Science and Technology, 42, 5131-5136.
European Medicines Agency (2004), Guideline on Environmental Impact Assessment for Veterinary Medicinal Products: Phase II, European Medicines Agency, London; available online http://www.emea.europa.eu/pdfs/vet/vich/079003en.pdf
Göbel, A.; McArdell, C. S.; Suter, M. J.-F.; Giger, W. (2004), ‘Trace determination of macrolide and sulfonamide antimicrobials, a human sulfonamide metabolite, and trimethoprim in wastewater using liquid chromatography coupled to electrospray tandem mass spectrometry’, Analytical Chemistry, 76, 4756-4764.
Goodyear, K., (2008), Sales of Antimicrobial Products Authorised for Use as Veterinary Medicines, Antiprotozoals, Antifungals, Growth Promoters and Coccidiostats, in the UK in 2007, Veterinary Medicines Directorate, Surrey.
Halling-Sørensen, B.; Nors Nielson, S.; Lanzky, P. F.; Ingerslev, F.; Holten Lützhøft, H. C.; Jørgensen, S. E. (1998), ‘Occurrence, fate and effects of pharmaceutical substances in the environment: A review’, Chemosphere, 36, 357-393.
Heberer, T. (2002), ‘Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: A review of recent research data’, Toxicology Letters, 131, 5-17.
Ingerslev, F.; Toräng, L.; Loke, M. L.; Halling-Sørensen, B.; Nyholm, N. (2001), ‘Primary biodegradation of veterinary antibiotics in aerobic and anaerobic surface water simulation systems’, Chemosphere, 44, 865-872.
Kay, P., Blackwell, P. A., Boxall, A. B. A. (2004), ‘Fate of veterinary antibiotics in a macroporous tile drained clay soil’, Environmental Toxicology and Chemistry, 23, 1136-1144.
Kay, P.; Blackwell, P. A.; Boxall, A. B. A. (2005a), ‘A lysimeter experiment to investigate the leaching of veterinary antibiotics through a clay soil and comparison with field data’, Environmental Pollution, 134, 333-341.
Kay, P.; Blackwell, P. A.; Boxall, A. B. A. (2005b), ‘Column studies to investigate the fate of veterinary antibiotics in clay soils following slurry application to agricultural land’, Chemosphere, 60, 497-507.
Kay, P.; Blackwell, P. A.; Boxall, A. B. A. (2005c), ‘Transport of veterinary antibiotics in overland flow following the application of slurry to arable land’, Chemosphere, 59, 951-959.
Kim, S-C.; Carlson, K. (2005), ‘LC-MS2 for quantifying trace amounts of pharmaceutical compounds in soil and sediment matrices’, Trends in Analytical Chemistry, 24, 635-644.
Knapp, C. W.; Engemann, C. A.; Hanson, M. L.; Keen, P. L.; Hall, K. J.; Graham, D. W. (2008), ‘Indirect evidence of transposon-mediated selection of antibiotic resistance genes in aquatic systems at low-level oxytetracycline exposures’, Environmental Science and Technology, 42, 5348-5353.
Kong, W.-D.; Zhu, Y.-G.; Fu, B.-J.; Marschner, P.; He, J.-Z. (2006), ‘The veterinary antibiotic oxytetracycline and Cu influence functional diversity of the soil microbial community’, Environmental Pollution, 143, 129-137.
Kong, W.-D.; Zhu, Y.-G.; Liang, Y. C.; Zhang, J.; Smith, F. A.; Yang, M. (2007), ‘Uptake of oxytetracycline and its phytotoxicity to alfalfa (Medicago sativa L.)’, Environmental Pollution, 147, 187-193.
Koschorreck, J.; Koch, C.; Rönnefahrt, I. (2002). ‘Environmental risk assessment of veterinary medicinal products in the EU: A regulatory perspective’, Toxicology Letters, 131, 117-124.
Lai, H.-T.; Liu, S.-M.; Chien, Y.-H. (1995), ‘Transformation of chloramphenicol and oxytetracycline in aquaculture pond sediments’, Journal of Environmental Science and Health: Part A, 30, 1897-1923.
Lindberg, R.; Jarnheimer, P.-A.; Olsen, B.; Johansson, M.; Tysklind, M. (2004). ‘Determination of antibiotic substances in hospital sewage water using solid phase extraction and liquid chromatography/mass spectrometry and group analogue internal standards’, Chemosphere, 57, 1479-1488.
McCracken, D. I. (1993), ‘The potential for avermectins to affect wildlife’, Veterinary Parasitology, 48, 273-280.
McKellar, Q. A. (1997), ‘Ecotoxicology and residues of anthelmintic compounds’, Veterinary Parasitology, 72, 413-435.
Nash, J. P.; Kime, D. E.; Van der Ven, L. T. M.; Wester, P. W.; Brion, F.; Maack, G.; Stahlschmidt-Allner, P.; Tyler, C. R. (2004), ‘Long-term exposure to environmental concentrations of the pharmaceutical ethynylestradiol causes reproductive failure in fish’, Environmental Health Perspectives, 112, 1725-1733.
Pérez-Lozano, P.; García-Montoya, E.; Orriols, A.; Miñarro, M.; Ticó, J .R.; Suñé-Negre, J. M. (2006), ‘Stability evaluation of amoxicillin in a solid premix veterinary formulation by monitoring the degradation products through a new HPLC analytical method’, Journal of Pharmaceutical and Biomedical Analysis, 42, 192-199.
Pruden, A.; Pei, R.; Storteboom, H.; Carlson, K. H. (2006), ‘Antibiotic resistance genes as emerging contaminants: Studies in Northern Colorado’, Environmental Science and Technology, 40, 7445-7450.
Rabølle, M.; Spliid, N. H. (2000), ‘Sorption and mobility of metronidazole, olaquindox, oxytetracycline and tylosin in soil’, Chemosphere, 40, 715-722.
Ridsdill-Smith, T. J. (1988), ‘Survival and reproduction of Musca vetustissima Walker (Dipteral Muscidae) and a scarabaeine dung beetle in dung cattle treated with avermectin B1’, Journal of the Australian Entomological Society, 27, 175-178.
Riediker, S.; Rytz, A.; Stadler, R. H. (2004), ‘Cold-temperature stability of five ß-lactam antibiotics in bovine milk and milk extracts prepared for liquid chromatography-electrospray ionization tandem mass spectrometry analysis’, Journal of Chromatography A, 1054, 359-363.
Russell, A. D. (2004), ‘Types of antibiotics and synthetic antimicrobial agents’, In Hugo & Russell's Pharmaceutical Microbiology, 7th edn., Denyer, S. P.; Hodges, N. A.; Gorman, S. P. (eds.), pp 152-186, Blackwell Science, Oxford.
Schreiber, F.; Szewzyk, U. (2008), ‘Environmentally relevant concentrations of pharmaceuticals influence the initial adhesion of bacteria’, Aquatic Toxicology, 87, 227-233.
Smith, A. (2004), ‘Bacterial resistance to antibiotics’, In Hugo & Russell's Pharmaceutical Microbiology, 7th edn., Denyer, S. P.; Hodges, N. A.; Gorman, S. P. (eds.), pp 220-232, Blackwell Science, Oxford.
Strong, L. (1993), ‘Overview: The impact of avermectins on pastureland ecology’, Veterinary Parasitology, 48, 3-17.
Svendsen, T. S.; Hansen, P. E.; Sommer, C.; Martinussen, T.; Grønvold, J.; Holter, P. (2005), ‘Life history characteristics of Lumbricus terrestris and effects of the veterinary antiparasitic compounds ivermectin and fenbendazole’, Soil Biology and Biochemistry, 37, 927-936.
The European Agency for the Evaluation of Medicinal Products (1999), Committee for Veterinary Medicinal Products: Ceftiofur Summary Report (2), EMEA/MRL/498/98.
Thiele-Bruhn, S.; Beck, I.-C. (2005), ‘Effects of sulfonamide and tetracycline antibiotics on soil microbial activity and microbial biomass’, Chemosphere, 59, 457-465.
Tolls, J. (2001), ‘Sorption of veterinary pharmaceuticals in soils: A review’, Environmental Science and Technology, 35, 3397-3406.
Wall, R.; Strong, L. (1987), ‘Environmental consequences of treating cattle with the antiparasitic drug ivermectin’, Nature, 327, 418-421.
Wollenberger, L.; Halling-Sørensen, B.; Kusk, K. O. (2000), ‘Acute and chronic toxicity of veterinary antibiotics to Daphnia magna’, Chemosphere, 40, 723-730.
MELANIE J. BOTTOMS
Department of Soil Science,
School of Human and Environmental Sciences,
University of Reading,
Whiteknights, Reading RG6 6DW
BIOGRAPHICAL NOTE: Melanie J. Bottoms graduated with a BSc (Hons) in Maritime Environmental Management from Southampton Solent University in 1998. She completed her MSc in Water Science at Lancaster University in 2004 and is currently (2009) in her final year of a PhD in Soil Science at the University of Reading. This article provides a brief summation of work already completed by other workers, providing a context for Melanie’s doctoral work.
[S1]The reference used the American tonnes rather than the English tons
It is clear that further investigation of the environmental fate and effects of amoxicillin is required, but additionally the other β-lactams should not be overlooked. In the UK in 2007, only 6 tonnes[S1] of active antimicrobial ingredient of cephalosporins were sold, but this is increasing year on year and has nearly doubled since 2004 (3,240 kg in 2004 compared to 6,215 kg in 2007) (Goodyear, 2008). In the case of ceftiofur, a cephalosporin antibiotic, it is known that more than 95% of the dose is excreted within twenty-four hours of administration in the urine and faeces as a combination of the parent compound and the two major metabolites (desfuroylceftiofur and desfuroylceftiofur cysteine disulphide) (The European Agency for the Evaluation of Medicinal Products, 1999). The two major metabolites retain the β-lactam moiety (The European Agency for the Evaluation of Medicinal Products, 1999), and thus retain the potential for biological activity.
Conclusions
The environmental fate of veterinary products is still a developing area of research. Work needs to proceed rapidly in order to enable policy makers to act with the most robust scientific evidence to protect both human health, and our environmental legacy for future generations. In particular, research to determine the ecotoxicological effects of the β-lactam drugs on key terrestrial indicator species would help establish the high priority substances from an environmental perspective. Additionally, characterisation of the major degradation products of the high use veterinary antibiotics is required. This is especially the case for the β-lactams, which are prone to hydrolysis in the environment (Cha et al., 2006), as there is little knowledge regarding the properties of β-lactam degradation products.
Acknowledgements
The author would like to thank her funding bodies, the EPSRC and the University of Reading’s Research Endowment Trust Fund. She would also like to thanks her supervisors Dr. Mark E. Hodson, Dr. Andrew Parker and Prof. Rainer Cramer for their support.
References
Andreozzi, R.; Caprio, V.; Ciniglia, C.; De Champdoré, M.; Lo Giudice, R.; Marotta, R.; Zuccato, E. (2004), ‘Antibiotics in the environment: Occurrence in Italian STPs, fate, and preliminary assessment on algal toxicity of amoxicillin’, Environmental Science and Technology, 38, 6832-6838.
Ash, R. J.; Mauck, B.; Morgan, M. (2002), ‘Antibiotic resistance of gram-negative bacteria in rivers, United States’, Emerging Infectious Diseases, 8, 713-716.
Benito-Peña, E.; Partal-Rodera, A. I.; León-González, M. E.; Moreno-Bondi, M. C. (2006), ‘Evaluation of mixed mode solid phase extraction cartridges for the preconcentration of ß-lactam antibiotics in wastewater using liquid chromatography with UV-DAD detection’, Analytica Chimica Acta, 556, 415-422.
Binh, C. T. T.; Heuer, H.; Gomes, N. C. M.; Kotzerke, A.; Fulle, M.; Wilke, B.-M.; Schloter, M.; Smalla, K. (2007), ‘Short-term effects of amoxicillin on bacterial communities in manured soil’, FEMS Microbiology Ecology, 62, 290-302.
Boleas, S.; Alonso, C.; Pro, J.; Fernández, C.; Carbonell, G.; Tarazona, J. V. (2005), ‘Toxicity of the antimicrobial oxytetracycline to soil organisms in a multi-species-soil system (MS.3) and influence of manure co-addition’, Journal of Hazardous Materials, 122, 233-241.
Boxall, A. B. A.; Blackwell, P.; Cavallo, R.; Kay, P.; Tolls, J. (2002a), ‘The sorption and transport of a sulphonamide antibiotic in soil systems’, Toxicology Letters, 131, 19-28.
Boxall, A. B. A.; Fogg, L. A.; Blackwell, P. A.; Kay, P.; Pemberton, E. J. (2002b), ‘Review of veterinary medicines in the environment’, R&D Technical Report P6-012/8/TR, Cranfield Centre for EcoChemistry, Environment Agency, Bristol; available online http://publications.environment-agency.gov.uk/pdf/SP6-012-8-TR-e-p.pdf
Boxall, A. B. A.; Fogg, L. A.; Kay, P.; Blackwell, P. A.; Pemberton, E. J.; Croxford, A. (2003), ‘Prioritisation of veterinary medicines in the UK environment’, Toxicology Letters, 142, 207-218.
Boxall, A. B. A.; Fogg, L. A.; Kay, P.; Blackwell, P. A.; Pemberton, E. J.; Croxford, A. (2004), ‘Veterinary medicines in the environment’, Reviews of Environmental Contamination and Toxicology, 180, 1-91.
Boxall, A. B. A.; Johnson, P.; Smith, E. J.; Sinclair, C. J.; Stutt, E.; Levy, L. S. (2006), ‘Uptake of veterinary medicines from soils into plants’, Journal of Agricultural and Food Chemistry, 54, 2288-2297.
Campagnolo, E. R.; Johnson, K. R.; Karpati, A.; Rubin, C. S.; Kolpin, D. W.; Meyer, M. T.; Esteban, J. E.; Currier, R. W.; Smith, K.; Thu, K. M.; McGeehin, M. (2002), ‘Antimicrobial residues in animal waste and water resources proximal to large-scale swine and poultry feeding operations’, The Science of the Total Environment, 299, 89-95.
Capleton, A. C.; Courage, C.; Rumsby, P.; Holmes, P.; Stutt, E.; Boxall, A. B. A.; Levy, L.S. (2006), ‘Prioritising veterinary medicines according to their potential indirect human exposure and toxicity profile’, Toxicology Letters, 163, 213-223.
Castiglioni, S.; Bagnati, R.; Fanelli, R.; Pomati, F.; Calamari, D.; Zuccato, E. (2006), ‘Removal of pharmaceuticals in sewage treatment plants in Italy’, Environmental Science and Technology, 40, 357-363.
Cha, J. M.; Yang, S.; Carlson, K. H. (2006), ‘Trace determination of ß-lactam antibiotics in surface water and urban wastewater using liquid chromatography combined with electrospray tandem mass spectrometry’, Journal of Chromatography A, 1115, 46-57.
Chee-Sanford, J. C.; Aminov, R. I.; Krapac, I. J.; Garrigues-Jeanjean, N.; Mackie, R. I. (2001), ‘Occurrence and diversity of tetracycline resistance genes in lagoons and groundwater underlying two swine production facilities’, Applied and Environmental Microbiology, 67, 1494-1502.
Christian, T.; Schneider, R. J.; Färber, H. A.; Skutlarek, D.; Meyer, M. T.; Goldbach, H. E. (2003), ‘Determination of antibiotics residues in manure, soil, and surface waters’, Acta Hydrochimica et Hydrobiologica, 31, 34-44.
Deshpande, A. D.; Baheti, K. G.; Chatterjee, N. R. (2004), ‘Degradation of ß-lactam antibiotics’, Current Science, 87, 1684-1695.
Díaz-Cruz, M.; López de Alda, M. J.; Barceló, D. (2003), ‘Environmental behaviour and analysis of veterinary and human drugs in soils, sediments and sludge’, Trends in Analytical Chemistry, 22, 340-351.
Engemann, C. A.; Keen, P. L.; Knapp, C. W.; Hall, K. J.; Graham, D. W. (2008), ‘Fate of tetracycline resistance genes in aquatic systems: migration from the water column to peripheral biofilms’, Environmental Science and Technology, 42, 5131-5136.
European Medicines Agency (2004), Guideline on Environmental Impact Assessment for Veterinary Medicinal Products: Phase II, European Medicines Agency, London; available online http://www.emea.europa.eu/pdfs/vet/vich/079003en.pdf
Göbel, A.; McArdell, C. S.; Suter, M. J.-F.; Giger, W. (2004), ‘Trace determination of macrolide and sulfonamide antimicrobials, a human sulfonamide metabolite, and trimethoprim in wastewater using liquid chromatography coupled to electrospray tandem mass spectrometry’, Analytical Chemistry, 76, 4756-4764.
Goodyear, K., (2008), Sales of Antimicrobial Products Authorised for Use as Veterinary Medicines, Antiprotozoals, Antifungals, Growth Promoters and Coccidiostats, in the UK in 2007, Veterinary Medicines Directorate, Surrey.
Halling-Sørensen, B.; Nors Nielson, S.; Lanzky, P. F.; Ingerslev, F.; Holten Lützhøft, H. C.; Jørgensen, S. E. (1998), ‘Occurrence, fate and effects of pharmaceutical substances in the environment: A review’, Chemosphere, 36, 357-393.
Heberer, T. (2002), ‘Occurrence, fate, and removal of pharmaceutical residues in the aquatic environment: A review of recent research data’, Toxicology Letters, 131, 5-17.
Ingerslev, F.; Toräng, L.; Loke, M. L.; Halling-Sørensen, B.; Nyholm, N. (2001), ‘Primary biodegradation of veterinary antibiotics in aerobic and anaerobic surface water simulation systems’, Chemosphere, 44, 865-872.
Kay, P., Blackwell, P. A., Boxall, A. B. A. (2004), ‘Fate of veterinary antibiotics in a macroporous tile drained clay soil’, Environmental Toxicology and Chemistry, 23, 1136-1144.
Kay, P.; Blackwell, P. A.; Boxall, A. B. A. (2005a), ‘A lysimeter experiment to investigate the leaching of veterinary antibiotics through a clay soil and comparison with field data’, Environmental Pollution, 134, 333-341.
Kay, P.; Blackwell, P. A.; Boxall, A. B. A. (2005b), ‘Column studies to investigate the fate of veterinary antibiotics in clay soils following slurry application to agricultural land’, Chemosphere, 60, 497-507.
Kay, P.; Blackwell, P. A.; Boxall, A. B. A. (2005c), ‘Transport of veterinary antibiotics in overland flow following the application of slurry to arable land’, Chemosphere, 59, 951-959.
Kim, S-C.; Carlson, K. (2005), ‘LC-MS2 for quantifying trace amounts of pharmaceutical compounds in soil and sediment matrices’, Trends in Analytical Chemistry, 24, 635-644.
Knapp, C. W.; Engemann, C. A.; Hanson, M. L.; Keen, P. L.; Hall, K. J.; Graham, D. W. (2008), ‘Indirect evidence of transposon-mediated selection of antibiotic resistance genes in aquatic systems at low-level oxytetracycline exposures’, Environmental Science and Technology, 42, 5348-5353.
Kong, W.-D.; Zhu, Y.-G.; Fu, B.-J.; Marschner, P.; He, J.-Z. (2006), ‘The veterinary antibiotic oxytetracycline and Cu influence functional diversity of the soil microbial community’, Environmental Pollution, 143, 129-137.
Kong, W.-D.; Zhu, Y.-G.; Liang, Y. C.; Zhang, J.; Smith, F. A.; Yang, M. (2007), ‘Uptake of oxytetracycline and its phytotoxicity to alfalfa (Medicago sativa L.)’, Environmental Pollution, 147, 187-193.
Koschorreck, J.; Koch, C.; Rönnefahrt, I. (2002). ‘Environmental risk assessment of veterinary medicinal products in the EU: A regulatory perspective’, Toxicology Letters, 131, 117-124.
Lai, H.-T.; Liu, S.-M.; Chien, Y.-H. (1995), ‘Transformation of chloramphenicol and oxytetracycline in aquaculture pond sediments’, Journal of Environmental Science and Health: Part A, 30, 1897-1923.
Lindberg, R.; Jarnheimer, P.-A.; Olsen, B.; Johansson, M.; Tysklind, M. (2004). ‘Determination of antibiotic substances in hospital sewage water using solid phase extraction and liquid chromatography/mass spectrometry and group analogue internal standards’, Chemosphere, 57, 1479-1488.
McCracken, D. I. (1993), ‘The potential for avermectins to affect wildlife’, Veterinary Parasitology, 48, 273-280.
McKellar, Q. A. (1997), ‘Ecotoxicology and residues of anthelmintic compounds’, Veterinary Parasitology, 72, 413-435.
Nash, J. P.; Kime, D. E.; Van der Ven, L. T. M.; Wester, P. W.; Brion, F.; Maack, G.; Stahlschmidt-Allner, P.; Tyler, C. R. (2004), ‘Long-term exposure to environmental concentrations of the pharmaceutical ethynylestradiol causes reproductive failure in fish’, Environmental Health Perspectives, 112, 1725-1733.
Pérez-Lozano, P.; García-Montoya, E.; Orriols, A.; Miñarro, M.; Ticó, J .R.; Suñé-Negre, J. M. (2006), ‘Stability evaluation of amoxicillin in a solid premix veterinary formulation by monitoring the degradation products through a new HPLC analytical method’, Journal of Pharmaceutical and Biomedical Analysis, 42, 192-199.
Pruden, A.; Pei, R.; Storteboom, H.; Carlson, K. H. (2006), ‘Antibiotic resistance genes as emerging contaminants: Studies in Northern Colorado’, Environmental Science and Technology, 40, 7445-7450.
Rabølle, M.; Spliid, N. H. (2000), ‘Sorption and mobility of metronidazole, olaquindox, oxytetracycline and tylosin in soil’, Chemosphere, 40, 715-722.
Ridsdill-Smith, T. J. (1988), ‘Survival and reproduction of Musca vetustissima Walker (Dipteral Muscidae) and a scarabaeine dung beetle in dung cattle treated with avermectin B1’, Journal of the Australian Entomological Society, 27, 175-178.
Riediker, S.; Rytz, A.; Stadler, R. H. (2004), ‘Cold-temperature stability of five ß-lactam antibiotics in bovine milk and milk extracts prepared for liquid chromatography-electrospray ionization tandem mass spectrometry analysis’, Journal of Chromatography A, 1054, 359-363.
Russell, A. D. (2004), ‘Types of antibiotics and synthetic antimicrobial agents’, In Hugo & Russell's Pharmaceutical Microbiology, 7th edn., Denyer, S. P.; Hodges, N. A.; Gorman, S. P. (eds.), pp 152-186, Blackwell Science, Oxford.
Schreiber, F.; Szewzyk, U. (2008), ‘Environmentally relevant concentrations of pharmaceuticals influence the initial adhesion of bacteria’, Aquatic Toxicology, 87, 227-233.
Smith, A. (2004), ‘Bacterial resistance to antibiotics’, In Hugo & Russell's Pharmaceutical Microbiology, 7th edn., Denyer, S. P.; Hodges, N. A.; Gorman, S. P. (eds.), pp 220-232, Blackwell Science, Oxford.
Strong, L. (1993), ‘Overview: The impact of avermectins on pastureland ecology’, Veterinary Parasitology, 48, 3-17.
Svendsen, T. S.; Hansen, P. E.; Sommer, C.; Martinussen, T.; Grønvold, J.; Holter, P. (2005), ‘Life history characteristics of Lumbricus terrestris and effects of the veterinary antiparasitic compounds ivermectin and fenbendazole’, Soil Biology and Biochemistry, 37, 927-936.
The European Agency for the Evaluation of Medicinal Products (1999), Committee for Veterinary Medicinal Products: Ceftiofur Summary Report (2), EMEA/MRL/498/98.
Thiele-Bruhn, S.; Beck, I.-C. (2005), ‘Effects of sulfonamide and tetracycline antibiotics on soil microbial activity and microbial biomass’, Chemosphere, 59, 457-465.
Tolls, J. (2001), ‘Sorption of veterinary pharmaceuticals in soils: A review’, Environmental Science and Technology, 35, 3397-3406.
Wall, R.; Strong, L. (1987), ‘Environmental consequences of treating cattle with the antiparasitic drug ivermectin’, Nature, 327, 418-421.
Wollenberger, L.; Halling-Sørensen, B.; Kusk, K. O. (2000), ‘Acute and chronic toxicity of veterinary antibiotics to Daphnia magna’, Chemosphere, 40, 723-730.
MELANIE J. BOTTOMS
Department of Soil Science,
School of Human and Environmental Sciences,
University of Reading,
Whiteknights, Reading RG6 6DW
BIOGRAPHICAL NOTE: Melanie J. Bottoms graduated with a BSc (Hons) in Maritime Environmental Management from Southampton Solent University in 1998. She completed her MSc in Water Science at Lancaster University in 2004 and is currently (2009) in her final year of a PhD in Soil Science at the University of Reading. This article provides a brief summation of work already completed by other workers, providing a context for Melanie’s doctoral work.
[S1]The reference used the American tonnes rather than the English tons